Myc, Oncogenic Protein Translation, and the Role of Polyamines
- PMID: 29799508
- PMCID: PMC6024823
- DOI: 10.3390/medsci6020041
Myc, Oncogenic Protein Translation, and the Role of Polyamines
Abstract
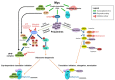
Deregulated protein synthesis is a common feature of cancer cells, with many oncogenic signaling pathways directly augmenting protein translation to support the biomass needs of proliferating tissues. MYC's ability to drive oncogenesis is a consequence of its essential role as a governor linking cell cycle entry with the requisite increase in protein synthetic capacity, among other biomass needs. To date, direct pharmacologic inhibition of MYC has proven difficult, but targeting oncogenic signaling modules downstream of MYC, such as the protein synthetic machinery, may provide a viable therapeutic strategy. Polyamines are essential cations found in nearly all living organisms that have both direct and indirect roles in the control of protein synthesis. Polyamine metabolism is coordinately regulated by MYC to increase polyamines in proliferative tissues, and this is further augmented in the many cancer cells harboring hyperactivated MYC. In this review, we discuss MYC-driven regulation of polyamines and protein synthetic capacity as a key function of its oncogenic output, and how this dependency may be perturbed through direct pharmacologic targeting of components of the protein synthetic machinery, such as the polyamines themselves, the eukaryotic translation initiation factor 4F (eIF4F) complex, and the eukaryotic translation initiation factor 5A (eIF5A).
Keywords: MYC; neuroblastoma; polyamines; protein synthesis in cancer.
Conflict of interest statement
The authors declare no conflict of interest. The funding sponsors had no role in the writing of or the decision to publish the manuscript.
Figures
References
-
- Hogarty M.D., Norris M.D., Davis K., Liu X., Evageliou N.F., Hayes C.S., Pawel B., Guo R., Zhao H., Sekyere E., et al. ODC1 is a critical determinant of MYCN oncogenesis and a therapeutic target in neuroblastoma. Cancer Res. 2008;68:9735–9745. doi: 10.1158/0008-5472.CAN-07-6866. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

