Association of an Opioid Standard of Practice Intervention With Intravenous Opioid Exposure in Hospitalized Patients
- PMID: 29799964
- PMCID: PMC6145746
- DOI: 10.1001/jamainternmed.2018.1044
Association of an Opioid Standard of Practice Intervention With Intravenous Opioid Exposure in Hospitalized Patients
Abstract
Importance: Opioids are commonly used to treat pain in hospitalized patients; however, intravenous administration carries an increased risk of adverse effects compared with oral administration. The subcutaneous route is an effective method of opioid delivery with favorable pharmacokinetics.
Objective: To assess an intervention to reduce intravenous opioid use, total parenteral opioid exposure, and the rate of patients administered parenteral opioids.
Design, setting, and participants: A pilot study was conducted in an adult general medical unit in an urban academic medical center. Attending physicians, nurse practitioners, and physician assistants who prescribed drugs were the participants. Use of opioids was compared between a 6-month control period and 3 months following education for the prescribers on opioid routes of administration.
Interventions: Adoption of a local opioid standard of practice, preferring the oral and subcutaneous routes over intravenous administration, and education for prescribers and nursing staff on awareness of the subcutaneous route was implemented.
Main outcomes and measures: The primary outcome was a reduction in intravenous doses administered per patient-day. Secondary measures included total parenteral and overall opioid doses per patient-day, parenteral and overall opioid exposure per patient-day, and daily rate of patients receiving parenteral opioids. Pain scores were measured on a standard 0- to 10-point Likert scale over the first 5 days of hospitalization.
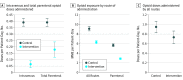
Results: The control period included 4500 patient-days, and the intervention period included 2459 patient-days. Of 127 patients in the intervention group, 59 (46.5%) were men; mean (SD) age was 57.6 (18.5) years. Intravenous opioid doses were reduced by 84% (0.06 vs 0.39 doses per patient-day, P < .001), and doses of all parenteral opioids were reduced by 55% (0.18 vs 0.39 doses per patient-day, P < .001). In addition, mean (SD) daily parenteral opioid exposure decreased by 49% (2.88 [0.72] vs 5.67 [1.14] morphine-milligram equivalents [MMEs] per patient-day). The daily rate of patients administered any parenteral opioid decreased by 57% (6% vs 14%; P < .001). Doses of opioids given by oral or parenteral route were reduced by 23% (0.73 vs 0.95 doses per patient-day, P = .02), and mean daily overall opioid exposure decreased by 31% (6.30 [4.12] vs 9.11 [7.34] MMEs per patient-day). For hospital days 1 through 3, there were no significant postintervention vs preintervention differences in mean reported pain score for patients receiving opioid therapy: day 1, -0.19 (95% CI, -0.94 to 0.56); day 2, -0.49 (95% CI, -1.01 to 0.03); and day 3, -0.54 (95% CI, -1.18 to 0.09). However, significant improvement was seen in the intervention group on days 4 (-1.07; 95% CI, -1.80 to -0.34) and 5 (-1.06; 95% CI, -1.84 to -0.27).
Conclusions and relevance: An intervention targeting the use of intravenous opioids may be associated with reduced opioid exposure while providing effective pain control to hospitalized adults.
Conflict of interest statement
Figures

References
-
- Radbruch L, Trottenberg P, Elsner F, Kaasa S, Caraceni A. Systematic review of the role of alternative application routes for opioid treatment for moderate to severe cancer pain: an EPCRC opioid guidelines project. Palliat Med. 2011;25(5):578-596. - PubMed
-
- Allain F, Minogianis E-A, Roberts DC, Samaha A-N. How fast and how often: the pharmacokinetics of drug use are decisive in addiction. Neurosci Biobehav Rev. 2015;56:166-179. - PubMed
-
- Waldmann CS, Eason JR, Rambohul E, Hanson GC. Serum morphine levels: a comparison between continuous subcutaneous infusion and continuous intravenous infusion in postoperative patients. Anaesthesia. 1984;39(8):768-771. - PubMed
-
- Munro AJ, Long GT, Sleigh JW. Nurse-administered subcutaneous morphine is a satisfactory alternative to intravenous patient-controlled analgesia morphine after cardiac surgery. Anesth Analg. 1998;87(1):11-15. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

