ER stress and unfolded protein response in ocular health and disease
- PMID: 29802807
- PMCID: PMC6583901
- DOI: 10.1111/febs.14522
ER stress and unfolded protein response in ocular health and disease
Abstract
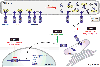
The human eye is the organ that is able to react to light in order to provide sharp three-dimensional and colored images. Unfortunately, the health of the eye can be impacted by various stimuli that can lead to vision loss, such as environmental changes, genetic mutations, or aging. Endoplasmic reticulum (ER) stress and unfolded protein response (UPR) signaling have been detected in many diverse ocular diseases, and chemical and genetic approaches to modulate ER stress and specific UPR regulatory molecules have shown beneficial effects in animal models of eye disease. This review highlights specific eye diseases associated with ER stress and UPR activity, based on a recent symposia exploring this theme.
Keywords: AMD; RGC; RPE; UPR; VEGF; ER stress; achromatopsia; glaucoma; neurodegernation; retinal degeneration.
Published 2018. This article is a U.S. Government work and is in the public domain in the USA.
Figures

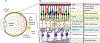
References
-
- Walter P & Ron D (2011) The unfolded protein response: from stress pathway to homeostatic regulation. Science 334, 1081–1086. - PubMed
-
- Shen J, Chen X, Hendershot L & Prywes R (2002) ER stress regulation of ATF6 localization by dissociation of BiP/GRP78 binding and unmasking of Golgi localization signals. Develop Cell 3, 99–111. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

