SAR studies on truxillic acid mono esters as a new class of antinociceptive agents targeting fatty acid binding proteins
- PMID: 29803996
- PMCID: PMC5999033
- DOI: 10.1016/j.ejmech.2018.04.050
SAR studies on truxillic acid mono esters as a new class of antinociceptive agents targeting fatty acid binding proteins
Abstract
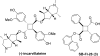
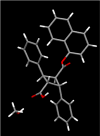
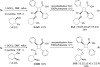
Fatty acid binding proteins (FABPs) serve as critical modulators of endocannabinoid signaling by facilitating the intracellular transport of anandamide and whose inhibition potentiates anandamide signaling. Our previous work has identified a novel small-molecule FABP inhibitor, α-truxillic acid 1-naphthyl monoester (SB-FI-26, 3) that has shown efficacy as an antinociceptive and anti-inflammatory agent in rodent models. In the present work, we have performed an extensive SAR study on a series of 3-analogs as novel FABP inhibitors based on computer-aided inhibitor drug design and docking analysis, chemical synthesis and biological evaluations. The prediction of binding affinity of these analogs to target FABP3, 5 and 7 isoforms was performed using the AutoDock 4.2 program, using the recently determined co-crystal structures of 3 with FABP5 and FABP7. The compounds with high docking scores were synthesized and evaluated for their activities using a fluorescence displacement assay against FABP3, 5 and 7. During lead optimization, compound 3l emerged as a promising compound with the Ki value of 0.21 μM for FABP 5, 4-fold more potent than 3 (Ki, 0.81 μM). Nine compounds exhibit similar or better binding affinity than 3, including compounds 4b (Ki, 0.55 μM) and 4e (Ki, 0.68 μM). Twelve compounds are selective for FABP5 and 7 with >10 μM Ki values for FABP3, indicating a safe profile to avoid potential cardiotoxicity concerns. Compounds 4f, 4j and 4k showed excellent selectivity for FABP5 and would serve as other new lead compounds. Compound 3a possessed high affinity and high selectivity for FABP7. Compounds with moderate to high affinity for FABP5 displayed antinociceptive effects in mice while compounds with low FABP5 affinity lacked in vivo efficacy. In vivo pain model studies in mice revealed that exceeding hydrophobicity significantly affects the efficacy. Thus, among the compounds with high affinity to FABP5 in vitro, the compounds with moderate hydrophobicity were identified as promising new lead compounds for the next round of optimization, including compounds 4b and 4j. For select cases, computational analysis of the observed SAR, especially the selectivity of new inhibitors to particular FABP isoforms, by comparing docking poses, interaction map, and docking energy scores has provided useful insights.
Keywords: Anti-inflammatory agent; Anti-nociceptive agent; Computer-aided design; FABP inhibitor; Fatty acid binding protein; Molecular docking; SAR study.
Copyright © 2018 Elsevier Masson SAS. All rights reserved.
Figures




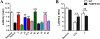
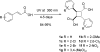
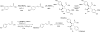


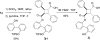



Similar articles
-
SAR study on Novel truxillic acid monoester-Based inhibitors of fatty acid binding proteins as Next-Generation antinociceptive agents.Bioorg Chem. 2022 Dec;129:106184. doi: 10.1016/j.bioorg.2022.106184. Epub 2022 Sep 30. Bioorg Chem. 2022. PMID: 36244323 Free PMC article.
-
The Antinociceptive Agent SBFI-26 Binds to Anandamide Transporters FABP5 and FABP7 at Two Different Sites.Biochemistry. 2017 Jul 11;56(27):3454-3462. doi: 10.1021/acs.biochem.7b00194. Epub 2017 Jun 28. Biochemistry. 2017. PMID: 28632393 Free PMC article.
-
Targeting fatty acid binding protein (FABP) anandamide transporters - a novel strategy for development of anti-inflammatory and anti-nociceptive drugs.PLoS One. 2012;7(12):e50968. doi: 10.1371/journal.pone.0050968. Epub 2012 Dec 7. PLoS One. 2012. PMID: 23236415 Free PMC article.
-
Potential safety implications of fatty acid-binding protein inhibition.Toxicol Appl Pharmacol. 2024 Oct;491:117079. doi: 10.1016/j.taap.2024.117079. Epub 2024 Aug 31. Toxicol Appl Pharmacol. 2024. PMID: 39218163 Review.
-
Small molecule inhibitors of human adipocyte fatty acid binding protein (FABP4).Med Chem. 2014 Jun;10(4):339-47. doi: 10.2174/15734064113096660045. Med Chem. 2014. PMID: 24024500 Review.
Cited by
-
Multiomic analysis of cervical squamous cell carcinoma identifies cellular ecosystems with biological and clinical relevance.Nat Genet. 2023 Dec;55(12):2175-2188. doi: 10.1038/s41588-023-01570-0. Epub 2023 Nov 20. Nat Genet. 2023. PMID: 37985817
-
Changes in median eminence of fatty acid-binding protein 3 in a mouse model of pain.Neuropsychopharmacol Rep. 2022 Mar;42(1):52-58. doi: 10.1002/npr2.12225. Epub 2022 Jan 28. Neuropsychopharmacol Rep. 2022. PMID: 35090101 Free PMC article.
-
Fatty acid binding protein 5 inhibitors as novel anticancer agents against metastatic castration-resistant prostate cancer.Bioorg Med Chem. 2025 May 1;122:118136. doi: 10.1016/j.bmc.2025.118136. Epub 2025 Feb 27. Bioorg Med Chem. 2025. PMID: 40058274
-
Identification of a novel fatty acid binding protein-5-CB2 receptor-dependent mechanism regulating anxiety behaviors in the prefrontal cortex.Cereb Cortex. 2023 Mar 10;33(6):2470-2484. doi: 10.1093/cercor/bhac220. Cereb Cortex. 2023. PMID: 35650684 Free PMC article.
-
FABP5 deletion in nociceptors augments endocannabinoid signaling and suppresses TRPV1 sensitization and inflammatory pain.Sci Rep. 2022 Jun 2;12(1):9241. doi: 10.1038/s41598-022-13284-0. Sci Rep. 2022. PMID: 35655086 Free PMC article. Clinical Trial.
References
-
- Lichtman AH, Shelton CC, Advani T, Cravatt BF. Mice lacking fatty acid amide hydrolase exhibit a cannabinoid receptor-mediated phenotypic hypoalgesia. Pain. 2004;109:319–327. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

