Baboon CD8 T cells suppress SIVmac infection in CD4 T cells through contact-dependent production of MIP-1α, MIP-1β, and RANTES
- PMID: 29807688
- PMCID: PMC6261791
- DOI: 10.1016/j.cyto.2018.05.022
Baboon CD8 T cells suppress SIVmac infection in CD4 T cells through contact-dependent production of MIP-1α, MIP-1β, and RANTES
Abstract
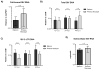
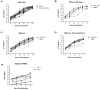
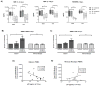
Simian immunodeficiency virus (SIV) infection in rhesus macaques is often characterized by high viremia and CD4 T cell depletion. By contrast, SIV infection in African nonhuman primate natural hosts is typically nonpathogenic despite active viral replication. Baboons are abundant in Africa and have a geographical distribution that overlaps with natural hosts, but they do not harbor SIVs. Previous work has demonstrated baboons are resistant to chronic SIV infection and/or disease in vivo but the underlying mechanisms remain unknown. Using in vitro SIVmac infections, we sought to identify SIV restriction factors in baboons by comparing observations to the pathogenic rhesus macaque model. SIVmac replicated in baboon PBMC but had delayed kinetics compared to rhesus PBMC. However, SIVmac replication in baboon and rhesus isolated CD4 cells were similar to the kinetics seen for rhesus PBMC, demonstrating intracellular restriction factors do not play a strong role in baboon inhibition of SIVmac replication. Here, we show CD8 T cells contribute to the innate SIV-suppressive activity seen in naïve baboon PBMC. As one mechanism of restriction, we identified higher production of MIP-1α, MIP-1β, and RANTES by baboon PBMC. Contact between CD4 and CD8 T cells resulted in maximum production of these chemokines and suppression of viral replication, whereas neutralization of CCR5-binding chemokines in baboon PBMC increased viral loads. Our studies indicate baboon natural restriction of SIVmac replication is largely dependent on CD4-extrinsinc mechanisms mediated, in part, by CD8 T cells.
Keywords: AIDS; Baboon; CCR5; Chemokine; Cytokine.
Copyright © 2018 Elsevier Ltd. All rights reserved.
Conflict of interest statement
None
Figures
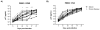

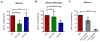

References
-
- Letvin NL, Daniel MD, Sehgal PK, Desrosiers RC, Hunt RD, Waldron LM, MacKey JJ, Schmidt DK, Chalifoux LV, King NW. Induction of AIDS-like disease in macaque monkeys with T-cell tropic retrovirus STLV-III. Science. 1985;230(4721):71–73. - PubMed
-
- Hahn BH, Shaw GM, De Cock KM, Sharp PM. AIDS as a zoonosis: scientific and public health implications. Science. 2000;287(5453):607–614. - PubMed
-
- Hausfater G. Predatory Behavior of Yellow Baboons. Behaviour. 1976;56(1/2):44–68.
-
- Kodama T, Silva DP, Daniel MD, Phillips-Conroy JE, Jolly CJ, Rogers J, Desrosiers RC. Prevalence of Antibodies to SIV in Baboons in Their Native Habitat. AIDS Res Hum Retroviruses. 1989;5(3):337–343. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

