Human Fibrinogen: Molecular and Genetic Aspects of Congenital Disorders
- PMID: 29844251
- PMCID: PMC6032319
- DOI: 10.3390/ijms19061597
Human Fibrinogen: Molecular and Genetic Aspects of Congenital Disorders
Abstract
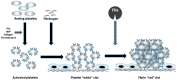
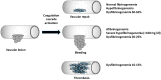
Congenital fibrinogen disorders can be quantitative (afibrinogenemia, hypofibrinogenemia) or functional (dysfibrinognemia). To date, several genetic variants have been identified in individuals with fibrinogen disorders. The complexity of the fibrinogen molecules, formed by three non-identical chains and with a trinodal organization, renders the identification of molecular causes and of clinical and biochemical phenotypes very challenging. However, the acknowledgement of the type of molecular defect is crucial for a safer therapy, which is going to improve the clinical management of these patients. In this review, some aspects concerning molecular and clinical findings available on congenital fibrinogen disorders will be discussed.
Keywords: afibrinogenemia; dysfibrinogenemia; hypofibrinogenemia.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Other Literature Sources

