E‑cadherin regulates proliferation of colorectal cancer stem cells through NANOG
- PMID: 29845283
- PMCID: PMC6072297
- DOI: 10.3892/or.2018.6464
E‑cadherin regulates proliferation of colorectal cancer stem cells through NANOG
Abstract
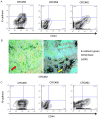
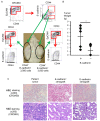
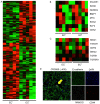
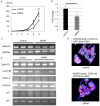
Cancer stem cells (CSCs) possess a self‑renewal ability and display tumorigenic potential in immunodeficient mice. Colorectal CSCs are thought to be a uniform population and no functionally distinct subpopulations have been identified. Because E‑cadherin is an essential molecule for self‑renewal of embryonic stem cells, we examined E‑cadherin expression, which may play a role in maintaining the properties of CSCs, in EpCAMhigh/CD44+ colorectal CSCs from human primary colorectal cancers. We obtained 18 surgical specimens of human primary colorectal cancer. CD44, EpCAM, and E‑cadherin expression were analyzed by fluorescence‑activated cell sorting. Sorted EpCAMhigh/CD44+ colorectal CSCs were injected into immunodeficient mice to estimate the tumorigenic potential. Genetic profiles were analyzed by cDNA microarray. Notably, colorectal CSCs could be divided into two populations based on the E‑cadherin expression status, and they exhibited different pathological characteristics. Compared to E‑cadherin‑negative colorectal CSCs, E‑cadherin‑positive (EC+) colorectal CSCs demonstrated higher tumor growth potential in vivo. EC+ colorectal CSCs revealed a higher expression of the pluripotency factor NANOG, which contributed to the higher tumor growth potential of EC+ colorectal CSCs through control of cyclin D1 expression. These findings are the first demonstration of functionally distinct subpopulations of colorectal CSCs in human clinical samples.
Figures

References
-
- Ishimoto T, Nagano O, Yae T, Tamada M, Motohara T, Oshima H, Oshima M, Ikeda T, Asaba R, Yagi H, et al. CD44 variant regulates redox status in cancer cells by stabilizing the xCT subunit of system xc- and thereby promotes tumor growth. Cancer Cell. 2011;19:387–400. doi: 10.1016/j.ccr.2011.01.038. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

