P2X7 ionotropic receptor is functionally expressed in rabbit articular chondrocytes and mediates extracellular ATP cytotoxicity
- PMID: 29845461
- PMCID: PMC6107462
- DOI: 10.1007/s11302-018-9611-x
P2X7 ionotropic receptor is functionally expressed in rabbit articular chondrocytes and mediates extracellular ATP cytotoxicity
Abstract
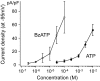
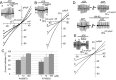
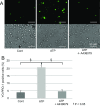
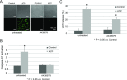
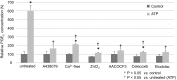
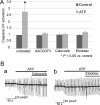
Extracellular ATP regulates various cellular functions by engaging multiple subtypes of P2 purinergic receptors. In many cell types, the ionotropic P2X7 receptor mediates pathological events such as inflammation and cell death. However, the importance of this receptor in chondrocytes remains largely unexplored. Here, we report the functional identification of P2X7 receptor in articular chondrocytes and investigate the involvement of P2X7 receptors in ATP-induced cytotoxicity. Chondrocytes were isolated from rabbit articular cartilage, and P2X7 receptor currents were examined using the whole-cell patch-clamp technique. ATP-induced cytotoxicity was evaluated by measuring caspase-3/7 activity, lactate dehydrogenase (LDH) leakage, and prostagrandin E2 (PGE2) release using microscopic and fluorimetric/colorimetric evaluation. Extracellular ATP readily evoked a cationic current without obvious desensitization. This ATP-activated current was dose related, but required millimolar concentrations. A more potent P2X7 receptor agonist, BzATP, also activated this current but at 100-fold lower concentrations. ATP-induced currents were largely abolished by selective P2X7 antagonists, suggesting a predominant role for the P2X7 receptor. RT-PCR confirmed the presence of P2X7 in chondrocytes. Heterologous expression of a rabbit P2X7 clone successfully reproduced the ATP-induced current. Exposure of chondrocytes to ATP increased caspase-3/7 activities, an effect that was totally abrogated by P2X7 receptor antagonists. Extracellular ATP also enhanced LDH release, which was partially attenuated by the P2X7 inhibitor. The P2X7 receptor-mediated elevation in apoptotic caspase signaling was accompanied by increased PGE2 release and was attenuated by inhibition of either phospholipase A2 or cyclooxygenase-2. This study provides direct evidence for the presence of functional P2X7 receptors in articular chondrocytes. Our results suggest that the P2X7 receptor is a potential therapeutic target in chondrocyte death associated with cartilage injury and disorders including osteoarthritis.
Keywords: ATP; Articular chondrocyte; Cytotoxicity; P2X7; Purinergic receptors.
Conflict of interest statement
Ethics
This study adheres to the ARRIVE guidelines for reporting animal research [55]. All the animal experiments were conducted in compliance with the protocol that was reviewed by the Institutional Animal Care and Use Committee and approved by the President of Shiga University of Medical Science (Permit Number: 2015-2-7).
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Figures



References
-
- Garcia M, Knight MM. Cyclic loading opens hemichannels to release ATP as part of a chondrocyte mechanotransduction pathway. J Orthop Res. 2010;28:510–515. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

