Extensive Bilateral Adrenal Rest Testicular Tumors in a Patient With 3 β-Hydroxysteroid Dehydrogenase Type 2 Deficiency
- PMID: 29850650
- PMCID: PMC5961024
- DOI: 10.1210/js.2018-00082
Extensive Bilateral Adrenal Rest Testicular Tumors in a Patient With 3 β-Hydroxysteroid Dehydrogenase Type 2 Deficiency
Abstract
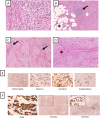
Testicular adrenal rest tumors (TARTs) are presumably derived from ectopic adrenocortical tissue in the testis, affecting up to 49% to 94% of males with congenital adrenal hyperplasia (CAH) due to 21-hydroxylase deficiency. Few reports have described TARTs in rarer forms of CAH such as 3β-hydroxysteroid dehydrogenase type 2 deficiency (3βHSD2D). A man with 3βHSD2D presented with massive bilateral testicular tumors. He had been treated with glucocorticoids and mineralocorticoids since infancy, with difficulties in suppressing dehydroepiandrosterone sulfate. At the age of 13 years, bilateral testicular lumps were found, and a radiologic diagnosis of TARTs was proposed. Subsequent sonographic examinations showed progression, despite intensifying his glucocorticoid therapy with metabolic complications. Following an open testicular biopsy, concerns of a Leydig cell tumor and risk of malignant transformation were raised, and because the patient also had local symptoms and azoospermia, he underwent bilateral orchiectomy at age 33 years. Histopathology was consistent with bilateral TARTs, exhibiting widespread immunoreactivity for adrenocortical markers, whereas no histological features of Leydig cell tumors were seen. The distinction between TARTs and Leydig cell tumors is important but can be challenging, and in our case, orchiectomy was needed to rule out the latter diagnosis. TART should be considered a differential diagnosis also in patients with 3βHSD2D who have testicular lumps.
Keywords: 3β-hydroxysteroid dehydrogenase deficiency; congenital adrenal hyperplasia; fertility; sperm quality; testicular adrenal rest tumors.
Figures
Similar articles
-
Clinical perspectives in congenital adrenal hyperplasia due to 3β-hydroxysteroid dehydrogenase type 2 deficiency.Endocrine. 2019 Mar;63(3):407-421. doi: 10.1007/s12020-018-01835-3. Epub 2019 Feb 4. Endocrine. 2019. PMID: 30719691 Free PMC article. Review.
-
Case Report:clinical experience of bilateral giant pediatric Testicular adrenal rest tumors with 3 Beta-Hydroxysteroid Dehydrogenase-2 family history.BMC Pediatr. 2021 Sep 15;21(1):405. doi: 10.1186/s12887-021-02883-x. BMC Pediatr. 2021. PMID: 34526000 Free PMC article.
-
Screening for testicular adrenal rest tumors among children with congenital adrenal hyperplasia at King Fahad Medical City, Saudi Arabia.J Pediatr Endocrinol Metab. 2021 Nov 9;35(1):49-54. doi: 10.1515/jpem-2021-0291. Print 2022 Jan 27. J Pediatr Endocrinol Metab. 2021. PMID: 34757702
-
Metachronous Bilateral Testicular Leydig-Like Tumors Leading to the Diagnosis of Congenital Adrenal Hyperplasia (Adrenogenital Syndrome).Case Rep Pathol. 2015;2015:459318. doi: 10.1155/2015/459318. Epub 2015 Aug 16. Case Rep Pathol. 2015. PMID: 26351608 Free PMC article.
-
Bilateral testicular masses and adrenal insufficiency: is congenital adrenal hyperplasia the only possible diagnosis? First two cases of TARTS described in Addison-only X-linked adrenoleukodystrophy and a brief review of literature.J Endocrinol Invest. 2021 Mar;44(3):391-402. doi: 10.1007/s40618-020-01362-x. Epub 2020 Jul 20. J Endocrinol Invest. 2021. PMID: 32691371 Review.
Cited by
-
Evaluation of 3β-hydroxysteroid dehydrogenase activity using progesterone and androgen receptors-mediated transactivation.Front Endocrinol (Lausanne). 2024 Oct 2;15:1480722. doi: 10.3389/fendo.2024.1480722. eCollection 2024. Front Endocrinol (Lausanne). 2024. PMID: 39415787 Free PMC article.
-
Approach of Heterogeneous Spectrum Involving 3beta-Hydroxysteroid Dehydrogenase 2 Deficiency.Diagnostics (Basel). 2022 Sep 7;12(9):2168. doi: 10.3390/diagnostics12092168. Diagnostics (Basel). 2022. PMID: 36140569 Free PMC article. Review.
-
Congenital Adrenal Hyperplasia-Current Insights in Pathophysiology, Diagnostics, and Management.Endocr Rev. 2022 Jan 12;43(1):91-159. doi: 10.1210/endrev/bnab016. Endocr Rev. 2022. PMID: 33961029 Free PMC article. Review.
-
Clinical perspectives in congenital adrenal hyperplasia due to 3β-hydroxysteroid dehydrogenase type 2 deficiency.Endocrine. 2019 Mar;63(3):407-421. doi: 10.1007/s12020-018-01835-3. Epub 2019 Feb 4. Endocrine. 2019. PMID: 30719691 Free PMC article. Review.
-
Genotype, Mortality, Morbidity, and Outcomes of 3β-Hydroxysteroid Dehydrogenase Deficiency in Algeria.Front Endocrinol (Lausanne). 2022 Jun 10;13:867073. doi: 10.3389/fendo.2022.867073. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35757411 Free PMC article.
References
-
- El-Maouche D, Arlt W, Merke DP. Congenital adrenal hyperplasia. Lancet. 2017;390(10108):2194–2210. - PubMed
-
- Falhammar H, Thorén M. Clinical outcomes in the management of congenital adrenal hyperplasia. Endocrine. 2012;41(3):355–373. - PubMed
-
- Stikkelbroeck NM, Otten BJ, Pasic A, Jager GJ, Sweep CG, Noordam K, Hermus AR. High prevalence of testicular adrenal rest tumors, impaired spermatogenesis, and Leydig cell failure in adolescent and adult males with congenital adrenal hyperplasia. J Clin Endocrinol Metab. 2001;86(12):5721–5728. - PubMed
-
- Engels M, Gehrmann K, Falhammar H, Webb EA, Nordenström A, Sweep FC, Span PN, van Herwaarden AE, Rohayem J, Richter-Unruh A, Bouvattier C, Köhler B, Kortmann BB, Arlt W, Roeleveld N, Reisch N, Stikkelbroeck NMML, Claahsen-van der Grinten HL; dsd-LIFE group . Gonadal function in adult male patients with congenital adrenal hyperplasia. Eur J Endocrinol. 2018;178(3):285–294. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
