Detection of Adverse Drug Reactions using Medical Named Entities on Twitter
- PMID: 29854190
- PMCID: PMC5977585
Detection of Adverse Drug Reactions using Medical Named Entities on Twitter
Abstract
Adverse Drug Reactions (ADRs) are unintentional reactions caused by a drug or combination of drugs taken by a patient. The current ADR reporting systems inevitably have delays in reporting such events. The broad scope of social media conversations on sites such as Twitter means that inevitably health-related topics will be covered. This means that these sites could then be used to detect potentially novel ADRs with less latency for subsequent further investigation. In this work, we investigate ADR surveillance using a large corpus of Twitter data, containing around 50 billion tweets spanning 3 years (2012-2014), and evaluate against over 3000 drugs reported in the FAERS database. This is both a larger corpus and broader selection of drugs than previous work in the domain. We compare the ADRs identified using our method to the FDA Adverse Event Reporting System (FAERS) database of ADRs reported using more traditional techniques, and find that Twitter is a useful resource for ADR detection up to 72% micro-averaged precision. Micro-averaged recall of 6% is achievable using only 10% of Twitter, indicating that with a higher-volume or targeted feed it would be possible to detect a large percentage of ADRs.
Figures
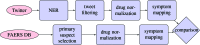
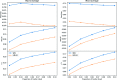
Similar articles
-
Complementing the US Food and Drug Administration Adverse Event Reporting System With Adverse Drug Reaction Reporting From Social Media: Comparative Analysis.JMIR Public Health Surveill. 2020 Sep 30;6(3):e19266. doi: 10.2196/19266. JMIR Public Health Surveill. 2020. PMID: 32996889 Free PMC article.
-
Combining Social Media and FDA Adverse Event Reporting System to Detect Adverse Drug Reactions.Drug Saf. 2020 Sep;43(9):893-903. doi: 10.1007/s40264-020-00943-2. Drug Saf. 2020. PMID: 32385840 Free PMC article.
-
Methods to Compare Adverse Events in Twitter to FAERS, Drug Information Databases, and Systematic Reviews: Proof of Concept with Adalimumab.Drug Saf. 2018 Dec;41(12):1397-1410. doi: 10.1007/s40264-018-0707-6. Drug Saf. 2018. PMID: 30167992 Free PMC article.
-
Analysis of Spontaneous Postmarket Case Reports Submitted to the FDA Regarding Thromboembolic Adverse Events and JAK Inhibitors.Drug Saf. 2018 Apr;41(4):357-361. doi: 10.1007/s40264-017-0622-2. Drug Saf. 2018. PMID: 29196988 Review.
-
Patients' role in reporting adverse drug reactions.Expert Opin Drug Saf. 2004 Jul;3(4):363-8. doi: 10.1517/14740338.3.4.363. Expert Opin Drug Saf. 2004. PMID: 15268652 Review.
Cited by
-
The Value of Social Media Analysis for Adverse Events Detection and Pharmacovigilance: Scoping Review.JMIR Public Health Surveill. 2024 Sep 6;10:e59167. doi: 10.2196/59167. JMIR Public Health Surveill. 2024. PMID: 39240684 Free PMC article.
-
A Disease Identification Algorithm for Medical Crowdfunding Campaigns: Validation Study.J Med Internet Res. 2022 Jun 21;24(6):e32867. doi: 10.2196/32867. J Med Internet Res. 2022. PMID: 35727610 Free PMC article.
-
Combining text mining with clinical decision support in clinical practice: a scoping review.J Am Med Inform Assoc. 2023 Feb 16;30(3):588-603. doi: 10.1093/jamia/ocac240. J Am Med Inform Assoc. 2023. PMID: 36512578 Free PMC article.
-
Complementing the US Food and Drug Administration Adverse Event Reporting System With Adverse Drug Reaction Reporting From Social Media: Comparative Analysis.JMIR Public Health Surveill. 2020 Sep 30;6(3):e19266. doi: 10.2196/19266. JMIR Public Health Surveill. 2020. PMID: 32996889 Free PMC article.
-
Combining Social Media and FDA Adverse Event Reporting System to Detect Adverse Drug Reactions.Drug Saf. 2020 Sep;43(9):893-903. doi: 10.1007/s40264-020-00943-2. Drug Saf. 2020. PMID: 32385840 Free PMC article.
References
-
- Shepherd Greene, Mohom Philip, Yacoub Kristina, Williams May Dianne. Adverse drug reaction deaths reported in United States vital statistics, 1999-2006. Annals of Pharmacotherapy. 2012;46(2):169–175. - PubMed
-
- National Bureau of Economic Research. Fda adverse event reporting system data – faers data. 2016. [Online; accessed 29-August-2016].
-
- Ma Paul, Marinovic Iván, Karaca-Mandic Pinar. Drug manufacturers delayed disclosure of serious and unexpected adverse events to the us food and drug administration. JAMA internal medicine. 2015;175(9):1565–1566. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
