Cx43 Channel Gating and Permeation: Multiple Phosphorylation-Dependent Roles of the Carboxyl Terminus
- PMID: 29867029
- PMCID: PMC6032060
- DOI: 10.3390/ijms19061659
Cx43 Channel Gating and Permeation: Multiple Phosphorylation-Dependent Roles of the Carboxyl Terminus
Abstract
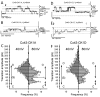
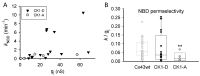
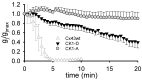
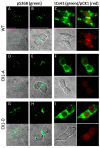
Connexin 43 (Cx43), a gap junction protein seemingly fit to support cardiac impulse propagation and synchronic contraction, is phosphorylated in normoxia by casein kinase 1 (CK1). However, during cardiac ischemia or pressure overload hypertrophy, this phosphorylation fades, Cx43 abundance decreases at intercalated disks and increases at myocytes' lateral borders, and the risk of arrhythmia rises. Studies in wild-type and transgenic mice indicate that enhanced CK1-phosphorylation of Cx43 protects from arrhythmia, while dephosphorylation precedes arrhythmia vulnerability. The mechanistic bases of these Cx43 (de)phosphoform-linked cardiac phenotypes are unknown. We used patch-clamp and dye injection techniques to study the channel function (gating, permeability) of Cx43 mutants wherein CK1-targeted serines were replaced by aspartate (Cx43-CK1-D) or alanine (Cx43-CK1-A) to emulate phosphorylation and dephosphorylation, respectively. Cx43-CK1-D, but not Cx43-CK1-A, displayed high Voltage-sensitivity and variable permselectivity. Both mutants showed multiple channel open states with overall increased conductivity, resistance to acidification-induced junctional uncoupling, and hemichannel openings in normal external calcium. Modest differences in the mutant channels' function and regulation imply the involvement of dissimilar structural conformations of the interacting domains of Cx43 in electrical and chemical gating that may contribute to the divergent phenotypes of CK1-(de)phospho-mimicking Cx43 transgenic mice and that may bear significance in arrhythmogenesis.
Keywords: arrhythmia; casein kinase 1; channel gating; gap junction permeability; phosphorylation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Channel Behavior and Voltage Gating of a Cx43 Mutant Simulating Preconditioning.Bioelectricity. 2023 Sep 1;5(3):181-187. doi: 10.1089/bioe.2023.0024. Epub 2023 Sep 12. Bioelectricity. 2023. PMID: 37746309 Free PMC article.
-
Connexin 43 phosphorylation by casein kinase 1 is essential for the cardioprotection by ischemic preconditioning.Basic Res Cardiol. 2021 Mar 22;116(1):21. doi: 10.1007/s00395-021-00861-z. Basic Res Cardiol. 2021. PMID: 33751227 Free PMC article.
-
Casein kinase 1 regulates connexin-43 gap junction assembly.J Biol Chem. 2002 Nov 22;277(47):44962-8. doi: 10.1074/jbc.M209427200. Epub 2002 Sep 20. J Biol Chem. 2002. PMID: 12270943
-
Emerging issues of connexin channels: biophysics fills the gap.Q Rev Biophys. 2001 Aug;34(3):325-472. doi: 10.1017/s0033583501003705. Q Rev Biophys. 2001. PMID: 11838236 Review.
-
The connexin 43 C-terminus: A tail of many tales.Biochim Biophys Acta Biomembr. 2018 Jan;1860(1):48-64. doi: 10.1016/j.bbamem.2017.05.008. Epub 2017 May 16. Biochim Biophys Acta Biomembr. 2018. PMID: 28526583 Review.
Cited by
-
Connexin43 in Musculoskeletal System: New Targets for Development and Disease Progression.Aging Dis. 2022 Dec 1;13(6):1715-1732. doi: 10.14336/AD.2022.0421. eCollection 2022 Dec 1. Aging Dis. 2022. PMID: 36465186 Free PMC article.
-
Novel Antidepressant Mechanism of Ginsenoside Rg1 in Regulating the Dysfunction of the Glutamatergic System in Astrocytes.Int J Mol Sci. 2022 Dec 29;24(1):575. doi: 10.3390/ijms24010575. Int J Mol Sci. 2022. PMID: 36614017 Free PMC article.
-
Abnormal Expression of Connexin43 in Cardiac Injury Induced by S-Band and X-Band Microwave Exposure in Rats.J Immunol Res. 2021 Dec 17;2021:3985697. doi: 10.1155/2021/3985697. eCollection 2021. J Immunol Res. 2021. PMID: 34957312 Free PMC article.
-
Astrocyte Networks as Therapeutic Targets in Glaucomatous Neurodegeneration.Cells. 2021 Jun 2;10(6):1368. doi: 10.3390/cells10061368. Cells. 2021. PMID: 34199470 Free PMC article. Review.
-
The Multifaceted Role of Astrocyte Connexin 43 in Ischemic Stroke Through Forming Hemichannels and Gap Junctions.Front Neurol. 2020 Jul 31;11:703. doi: 10.3389/fneur.2020.00703. eCollection 2020. Front Neurol. 2020. PMID: 32849190 Free PMC article. Review.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

