Mifepristone Pretreatment for the Medical Management of Early Pregnancy Loss
- PMID: 29874535
- PMCID: PMC6437668
- DOI: 10.1056/NEJMoa1715726
Mifepristone Pretreatment for the Medical Management of Early Pregnancy Loss
Abstract
Background: Medical management of early pregnancy loss is an alternative to uterine aspiration, but standard medical treatment with misoprostol commonly results in treatment failure. We compared the efficacy and safety of pretreatment with mifepristone followed by treatment with misoprostol with the efficacy and safety of misoprostol use alone for the management of early pregnancy loss.
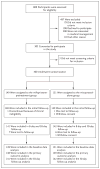
Methods: We randomly assigned 300 women who had an anembryonic gestation or in whom embryonic or fetal death was confirmed to receive pretreatment with 200 mg of mifepristone, administered orally, followed by 800 μg of misoprostol, administered vaginally (mifepristone-pretreatment group), or 800 μg of misoprostol alone, administered vaginally (misoprostol-alone group). Participants returned 1 to 4 days after misoprostol use for evaluation, including ultrasound examination, by an investigator who was unaware of the treatment-group assignments. Women in whom the gestational sac was not expelled were offered expectant management, a second dose of misoprostol, or uterine aspiration. We followed all participants for 30 days after randomization. Our primary outcome was gestational sac expulsion with one dose of misoprostol by the first follow-up visit and no additional intervention within 30 days after treatment.
Results: Complete expulsion after one dose of misoprostol occurred in 124 of 148 women (83.8%; 95% confidence interval [CI], 76.8 to 89.3) in the mifepristone-pretreatment group and in 100 of 149 women (67.1%; 95% CI, 59.0 to 74.6) in the misoprostol-alone group (relative risk, 1.25; 95% CI, 1.09 to 1.43). Uterine aspiration was performed less frequently in the mifepristone-pretreatment group than in the misoprostol-alone group (8.8% vs. 23.5%; relative risk, 0.37; 95% CI, 0.21 to 0.68). Bleeding that resulted in blood transfusion occurred in 2.0% of the women in the mifepristone-pretreatment group and in 0.7% of the women in the misoprostol-alone group (P=0.31); pelvic infection was diagnosed in 1.3% of the women in each group.
Conclusions: Pretreatment with mifepristone followed by treatment with misoprostol resulted in a higher likelihood of successful management of first-trimester pregnancy loss than treatment with misoprostol alone. (Funded by the National Institute of Child Health and Human Development; PreFaiR ClinicalTrials.gov number, NCT02012491 .).
Conflict of interest statement
Dr. Creinin reports receiving consulting fees from Danco Laboratories. No other potential conflict of interest relevant to this article was reported.
Figures

Comment in
-
A Better Medical Regimen for the Management of Miscarriage.N Engl J Med. 2018 Jun 7;378(23):2232-2233. doi: 10.1056/NEJMe1803491. N Engl J Med. 2018. PMID: 29874544 No abstract available.
-
Excerpts from the World Medical Literature.J Obstet Gynaecol Can. 2018 Nov;40(11):1387-1389. doi: 10.1016/j.jogc.2018.08.020. J Obstet Gynaecol Can. 2018. PMID: 30473115 No abstract available.
References
-
- Casterline JB. Collecting data on pregnancy loss: a review of evidence from the World Fertility Survey. Stud Fam Plann. 1989;20:81–95. - PubMed
-
- Ventura SJ, Curtin SC, Abma JC, Henshaw SK. Estimated pregnancy rates and rates of pregnancy outcomes for the United States, 1990–2008. Natl Vital Stat Rep. 2012;60:1–21. - PubMed
-
- Hertig AT, Livingstone RG. Spontaneous, threatened and habitual abortion: their pathogenesis and treatment. N Engl J Med. 1944;230:797–806.
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
