NAP enzyme recruitment in simultaneous bioremediation and nanoparticles synthesis
- PMID: 29876306
- PMCID: PMC5989592
- DOI: 10.1016/j.btre.2018.e00257
NAP enzyme recruitment in simultaneous bioremediation and nanoparticles synthesis
Erratum in
-
Erratum regarding missing Declaration of Competing Interest statements in previously published articles.Biotechnol Rep (Amst). 2021 Mar 19;29:e00582. doi: 10.1016/j.btre.2020.e00582. eCollection 2021 Mar. Biotechnol Rep (Amst). 2021. PMID: 33786326 Free PMC article.
Abstract
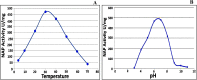
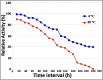
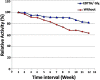
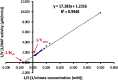
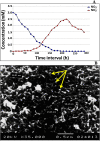
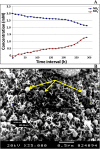
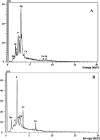
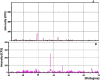
The periplasmic nitrate reductase enzyme (NAP) has become attractive catalyst, whose exploitation has emerged as one of the indispensable strategies toward environmentally benign applications. To achieve them efficiently and overcome the sensitivity of NAP in harsh environmental circumstances, the immobilization for denitrifying bacteria and NAP enzyme for simultaneous bioremediation and bionanoparticles synthesis was studied. NAP catalyzed NO3- reduction at Vmax of 0.811 μM/min and Km of 14.02 mM. Concurrently, the immobilized MMT cells completely removed NO3- upon 192 h with AgNPs synthesis ranging from 23.26 to 58.14 nm as indicated by SEM. Wherase, immobilized NAP exhibited lower efficiency with 28.6% of NO3- elimination within 288 h and large aggregated AgNPs ranging from 94.44 nm to 172.22 nm. To the best of author knowledge, the immobilization for denitrifying bacteria and NAP enzyme for simultaneous bioremediation and bionanoparticles synthesis was not studied before.
Keywords: Bioremediation; Denitrification; Immobilization; Nanoparticles; Nitrate reductase.
Figures

References
-
- Shrimali M., Singh K. New methods of nitrate removal from water$ Environ. Pollut. 2001;112:351–359. - PubMed
-
- Okolo J., Nweke C., Nwabueze R., Dike C., Nwanyanwu C. Toxicity of phenolic compounds to oxidoreductases of Acinetobacter species isolated from a tropical soil. Sci. Res. Essays. 2007;2:244–250.
-
- Morozkina E., Zvyagilskaya R. Nitrate reductases: structure, functions, and effect of stress factors. Biochemistry (Mosc.) 2007;72(10):1151–1160. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
