The role of G-protein-coupled membrane estrogen receptor in mouse Leydig cell function-in vivo and in vitro evaluation
- PMID: 29876633
- PMCID: PMC6209072
- DOI: 10.1007/s00441-018-2861-7
The role of G-protein-coupled membrane estrogen receptor in mouse Leydig cell function-in vivo and in vitro evaluation
Abstract
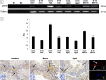
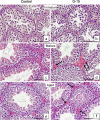
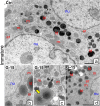
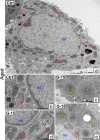
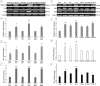
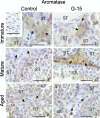
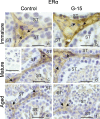
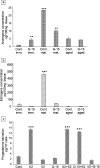
In this study, G-coupled estrogen receptor (GPER) was inactivated, by treatment with antagonist (G-15), in testes of C57BL/6 mice: immature (3 weeks old), mature (3 months old) and aged (1.5 years old) (50 μg/kg bw), as well as MA-10 mouse Leydig cells (10 nM/24 h) alone or in combination with 17β-estradiol or antiestrogen (ICI 182,780). In G-15-treated mice, overgrowth of interstitial tissue was found in both mature and aged testes. Depending on age, differences in structure and distribution of various Leydig cell organelles were observed. Concomitantly, modulation of activity of the mitochondria and tubulin microfibers was revealed. Diverse and complex GPER regulation at the mRNA level and protein of estrogen signaling molecules (estrogen receptor α and β; ERα, ERβ and cytochrome P450 aromatase; P450arom) in G-15 Leydig cells was found in relation to age and the experimental system utilized (in vivo and in vitro). Changes in expression patterns of ERs and P450arom, as well as steroid secretion, reflected Leydig cell heterogeneity to estrogen regulation throughout male life including cell physiological status.We show, for the first time, GPER with ERs and P450arom work in tandem to maintain Leydig cell architecture and supervise its steroidogenic function by estrogen during male life. Full set of estrogen signaling molecules, with involvement of GPER, is crucial for proper Leydig cell function where each molecule acts in a specific and/or complementary manner. Further understanding of the mechanisms by which GPER controls Leydig cells with special regard to male age, cell of origin and experimental system used is critical for predicting and preventing testis steroidogenic disorders based on perturbations in estrogen signaling.
Keywords: Estrogen receptors, estrogens; G-coupled estrogen receptor; Leydig cell; Ultrastructure.
Conflict of interest statement
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All applicable international, national and/or institutional guidelines for the care and use of animals were followed.
Informed consent
Not applicable.
Figures




References
-
- Abney TO. The potential roles of estrogens in regulating Leydig cell development and function: a review. Steroids. 1999;64(9):610–617. - PubMed
-
- Abney TO, Myers RB. 17 beta-estradiol inhibition of Leydig cell regeneration in the ethane dimethylsulfonate-treated mature rat. J Androl. 1991;12(5):295–304. - PubMed
-
- Abraham GE, Swerdloff ES, Tulchinsky D, Odell WD. Radioimmunoassay of plasma 17-hydroxyprogesterone. J Clin Endocrinol. 1971;33:42–46. - PubMed
-
- Acconcia Filippo, Fiocchetti Marco, Marino Maria. Xenoestrogen regulation of ERα/ERβ balance in hormone-associated cancers. Molecular and Cellular Endocrinology. 2017;457:3–12. - PubMed
-
- Alarid ET, Bakopoulos N, Solodin N. Proteasome-mediated proteolysis of estrogen receptor: a novel component in autologous down-regulation. Mol Endocrinol. 1999;13:1522–1534. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

