Role of Caenorhabditis elegans AKT-1/2 and SGK-1 in Manganese Toxicity
- PMID: 29882004
- PMCID: PMC6286235
- DOI: 10.1007/s12640-018-9915-1
Role of Caenorhabditis elegans AKT-1/2 and SGK-1 in Manganese Toxicity
Abstract
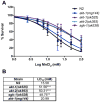
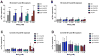
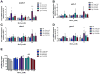
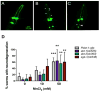
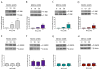
Excessive levels of the essential metal manganese (Mn) may cause a syndrome similar to Parkinson's disease. The model organism Caenorhabditis elegans mimics some of Mn effects in mammals, including dopaminergic neurodegeneration, oxidative stress, and increased levels of AKT. The evolutionarily conserved insulin/insulin-like growth factor-1 signaling pathway (IIS) modulates worm longevity, metabolism, and antioxidant responses by antagonizing the transcription factors DAF-16/FOXO and SKN-1/Nrf-2. AKT-1, AKT-2, and SGK-1 act upstream of these transcription factors. To study the role of these proteins in C. elegans response to Mn intoxication, wild-type N2 and loss-of-function mutants were exposed to Mn (2.5 to 100 mM) for 1 h at the L1 larval stage. Strains with loss-of-function in akt-1, akt-2, and sgk-1 had higher resistance to Mn compared to N2 in the survival test. All strains tested accumulated Mn similarly, as shown by ICP-MS. DAF-16 nuclear translocation was observed by fluorescence microscopy in WT and loss-of-function strains exposed to Mn. qRT-PCR data indicate increased expression of γ-glutamyl cysteine synthetase (GCS-1) antioxidant enzyme in akt-1 mutants. The expression of sod-3 (superoxide dismutase homologue) was increased in the akt-1 mutant worms, independent of Mn treatment. However, dopaminergic neurons degenerated even in the more resistant strains. Dopaminergic function was evaluated with the basal slowing response behavioral test and dopaminergic neuron integrity was evaluated using worms expressing green fluorescent protein (GFP) under the dopamine transporter (DAT-1) promoter. These results suggest that AKT-1/2 and SGK-1 play a role in C. elegans response to Mn intoxication. However, tissue-specific responses may occur in dopaminergic neurons, contributing to degeneration.
Keywords: Akt/PKB; C. elegans; DAF-16; Manganese; SGK-1; Signaling pathways.
Conflict of interest statement
Figures

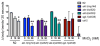

Similar articles
-
Null allele mutants of trt-1, the catalytic subunit of telomerase in Caenorhabditis elegans, are less sensitive to Mn-induced toxicity and DAergic degeneration.Neurotoxicology. 2016 Dec;57:54-60. doi: 10.1016/j.neuro.2016.08.016. Epub 2016 Sep 1. Neurotoxicology. 2016. PMID: 27593554
-
Effects of Caenorhabditis elegans sgk-1 mutations on lifespan, stress resistance, and DAF-16/FoxO regulation.Aging Cell. 2013 Oct;12(5):932-40. doi: 10.1111/acel.12120. Epub 2013 Jul 19. Aging Cell. 2013. PMID: 23786484 Free PMC article.
-
The Nrf2/SKN-1-dependent glutathione S-transferase π homologue GST-1 inhibits dopamine neuron degeneration in a Caenorhabditis elegans model of manganism.Neurotoxicology. 2013 Sep;38:51-60. doi: 10.1016/j.neuro.2013.05.014. Epub 2013 May 27. Neurotoxicology. 2013. PMID: 23721876 Free PMC article.
-
Altered manganese homeostasis: implications for BLI-3-dependent dopaminergic neurodegeneration and SKN-1 protection in C. elegans.J Trace Elem Med Biol. 2012 Jun;26(2-3):183-7. doi: 10.1016/j.jtemb.2012.03.011. Epub 2012 May 15. J Trace Elem Med Biol. 2012. PMID: 22591558 Review.
-
Caenorhabditis elegans as a Model to Study Manganese-Induced Neurotoxicity.Biomolecules. 2022 Sep 29;12(10):1396. doi: 10.3390/biom12101396. Biomolecules. 2022. PMID: 36291605 Free PMC article. Review.
Cited by
-
Modeling neurodegeneration in Caenorhabditiselegans.Dis Model Mech. 2020 Oct 26;13(10):dmm046110. doi: 10.1242/dmm.046110. Dis Model Mech. 2020. PMID: 33106318 Free PMC article. Review.
-
The impact of manganese on neurotransmitter systems.J Trace Elem Med Biol. 2020 Sep;61:126554. doi: 10.1016/j.jtemb.2020.126554. Epub 2020 May 20. J Trace Elem Med Biol. 2020. PMID: 32480053 Free PMC article. Review.
-
Limosilactobacillus fermentum MG4244 Protects Against Metabolic and Inflammatory Stress in Caenorhabditis elegans.Foods. 2025 Jun 5;14(11):1995. doi: 10.3390/foods14111995. Foods. 2025. PMID: 40509522 Free PMC article.
-
Domain definition and preliminary functional exploration of the endonuclease NOBP-1 in Strongyloides stercoralis.Parasit Vectors. 2023 Nov 3;16(1):399. doi: 10.1186/s13071-023-05940-9. Parasit Vectors. 2023. PMID: 37924155 Free PMC article.
-
In vivo pulse-chase in C. elegans reveals intestinal histone turnover changes upon starvation.bioRxiv [Preprint]. 2025 Feb 16:2025.02.13.638128. doi: 10.1101/2025.02.13.638128. bioRxiv. 2025. Update in: J Biol Chem. 2025 Jul;301(7):110299. doi: 10.1016/j.jbc.2025.110299. PMID: 39990428 Free PMC article. Updated. Preprint.
References
-
- Altun ZF, Hall DH. Nervous system, general description. [Accessed September 17 2016];WormAtlas. 2011
MeSH terms
Substances
Grants and funding
- 202662/2014-4/Conselho Nacional de Desenvolvimento Científico e Tecnológico
- R01 ES10563/National Institutes of Health
- P40 OD010440/OD/NIH HHS/United States
- R01 ES020852/ES/NIEHS NIH HHS/United States
- 4103/2-1/Deutsche Forschungsgemeinschaft
- 903/9-1/Deutsche Forschungsgemeinschaft
- 0407/13-5/Coordenação de Aperfeiçoamento de Pessoal de Nível Superior
- R01 ES07331/National Institutes of Health
- R01 ES020852/National Institutes of Health
- R01 ES007331/ES/NIEHS NIH HHS/United States
- P30 CA013330/CA/NCI NIH HHS/United States
- R01 ES010563/ES/NIEHS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

