Bovine respiratory syncytial virus infection enhances Pasteurella multocida adherence on respiratory epithelial cells
- PMID: 29885798
- PMCID: PMC7117154
- DOI: 10.1016/j.vetmic.2018.04.031
Bovine respiratory syncytial virus infection enhances Pasteurella multocida adherence on respiratory epithelial cells
Abstract
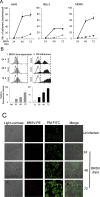
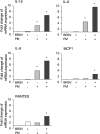
Primary infection with bovine respiratory syncytial virus (BRSV) predisposes cattle to secondary infection with bacteria that cause bovine respiratory disease complex (BRDC). However, the interaction between BRSV and bacteria is unclear. This in vitro study examined the adherence of Pasteurella multocida (PM) to BRSV-infected cells was assessed in colony forming unit assays, by flow cytometry analysis, and by indirect immunofluorescence analysis (IFA) of epithelial cells (A549, HEp-2, and MDBK). An in vitro model based on infection of BRSV-infected epithelial cells revealed that PM adherence to BRSV-infected cells was 2- to 8-fold higher than uninfected cells. This was confirmed by flow cytometry analysis and IFA. Epithelial cell expression of mRNA encoding cytokines and chemokines increased after exposure to PM, but increased further after co-infection with BRSV and PM. BRSV-mediated adherence of PM to epithelial cells may underlie the serious symptoms of BRDC.
Keywords: Adherent bacteria; BRDC; BRSV; Co-infection.
Copyright © 2018 Elsevier B.V. All rights reserved.
Figures

Similar articles
-
Bovine Respiratory Syncytial Virus Decreased Pasteurella multocida Adherence by Downregulating the Expression of Intercellular Adhesion Molecule-1 on the Surface of Upper Respiratory Epithelial Cells.Vet Microbiol. 2020 Jul;246:108748. doi: 10.1016/j.vetmic.2020.108748. Epub 2020 Jun 2. Vet Microbiol. 2020. PMID: 32605748 Free PMC article.
-
Bovine respiratory syncytial virus enhances the attachment of Trueperella pyogenes to cells.J Vet Med Sci. 2024 Oct 1;86(10):1068-1075. doi: 10.1292/jvms.24-0068. Epub 2024 Aug 8. J Vet Med Sci. 2024. PMID: 39111845 Free PMC article.
-
Co-infection of epithelial cells established from the upper and lower bovine respiratory tract with bovine respiratory syncytial virus and bacteria.Vet Microbiol. 2019 Aug;235:80-85. doi: 10.1016/j.vetmic.2019.06.010. Epub 2019 Jun 12. Vet Microbiol. 2019. PMID: 31282382
-
Bovine respiratory syncytial virus.Vet Clin North Am Food Anim Pract. 2010 Jul;26(2):323-33. doi: 10.1016/j.cvfa.2010.04.010. Vet Clin North Am Food Anim Pract. 2010. PMID: 20619187 Review.
-
Bovine respiratory syncytial virus infection: immunopathogenic mechanisms.Anim Health Res Rev. 2007 Dec;8(2):207-13. doi: 10.1017/S1466252307001405. Anim Health Res Rev. 2007. PMID: 18218161 Review.
Cited by
-
The Immunology of Bovine Respiratory Disease: Recent Advancements.Vet Clin North Am Food Anim Pract. 2020 Jul;36(2):333-348. doi: 10.1016/j.cvfa.2020.03.002. Epub 2020 Apr 21. Vet Clin North Am Food Anim Pract. 2020. PMID: 32327252 Free PMC article. Review.
-
Bovine Respiratory Syncytial Virus Decreased Pasteurella multocida Adherence by Downregulating the Expression of Intercellular Adhesion Molecule-1 on the Surface of Upper Respiratory Epithelial Cells.Vet Microbiol. 2020 Jul;246:108748. doi: 10.1016/j.vetmic.2020.108748. Epub 2020 Jun 2. Vet Microbiol. 2020. PMID: 32605748 Free PMC article.
-
Prevalence of BRD-Related Viral Pathogens in the Upper Respiratory Tract of Swiss Veal Calves.Animals (Basel). 2021 Jun 29;11(7):1940. doi: 10.3390/ani11071940. Animals (Basel). 2021. PMID: 34209718 Free PMC article.
-
Design of a High-Throughput Real-Time PCR System for Detection of Bovine Respiratory and Enteric Pathogens.Front Vet Sci. 2021 Jun 24;8:677993. doi: 10.3389/fvets.2021.677993. eCollection 2021. Front Vet Sci. 2021. PMID: 34250065 Free PMC article.
-
Bovine respiratory syncytial virus enhances the attachment of Trueperella pyogenes to cells.J Vet Med Sci. 2024 Oct 1;86(10):1068-1075. doi: 10.1292/jvms.24-0068. Epub 2024 Aug 8. J Vet Med Sci. 2024. PMID: 39111845 Free PMC article.
References
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

