Distinct Circuits for Recovery of Eye Dominance and Acuity in Murine Amblyopia
- PMID: 29887305
- PMCID: PMC6008222
- DOI: 10.1016/j.cub.2018.04.055
Distinct Circuits for Recovery of Eye Dominance and Acuity in Murine Amblyopia
Abstract
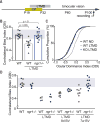
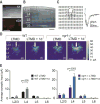
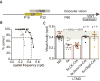
Degrading vision by one eye during a developmental critical period yields enduring deficits in both eye dominance and visual acuity. A predominant model is that "reactivating" ocular dominance (OD) plasticity after the critical period is required to improve acuity in amblyopic adults. However, here we demonstrate that plasticity of eye dominance and acuity are independent and restricted by the nogo-66 receptor (ngr1) in distinct neuronal populations. Ngr1 mutant mice display greater excitatory synaptic input onto both inhibitory and excitatory neurons with restoration of normal vision. Deleting ngr1 in excitatory cortical neurons permits recovery of eye dominance but not acuity. Reciprocally, deleting ngr1 in thalamus is insufficient to rectify eye dominance but yields improvement of acuity to normal. Abolishing ngr1 expression in adult mice also promotes recovery of acuity. Together, these findings challenge the notion that mechanisms for OD plasticity contribute to the alterations in circuitry that restore acuity in amblyopia.
Keywords: amblyopia; eye dominance; leucine-rich repeat; plasticity; visual acuity.
Copyright © 2018 Elsevier Ltd. All rights reserved.
Conflict of interest statement
The authors declare no competing interest
Figures
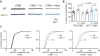


Comment in
-
Amblyopia: The Thalamus Is a No-Go Area for Visual Acuity.Curr Biol. 2018 Jun 18;28(12):R709-R712. doi: 10.1016/j.cub.2018.04.081. Curr Biol. 2018. PMID: 29920266
References
-
- Webber AL, Wood J. Amblyopia: prevalence, natural history, functional effects and treatment. Clin Exp Optometry. 2005;88:365–375. - PubMed
-
- Sengpiel F. Plasticity of the visual cortex and treatment of amblyopia. Curr Biol. 2014;24:R936–R940. - PubMed
-
- Kratz KE, Mangel SC, Lehmkuhle S, Sherman M. Retinal X- and Y-cells in monocularly lid-sutured cats: normality of spatial and temporal properties. Brain Res. 1979;172:545–551. - PubMed
-
- Kiorpes L. Visual processing in amblyopia: animal studies. Strabismus. 2006;14:3–10. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Research Materials

