Tolerance to Morphine-Induced Inhibition of TTX-R Sodium Channels in Dorsal Root Ganglia Neurons Is Modulated by Gut-Derived Mediators
- PMID: 29888757
- PMCID: PMC5993194
- DOI: 10.1016/j.isci.2018.03.003
Tolerance to Morphine-Induced Inhibition of TTX-R Sodium Channels in Dorsal Root Ganglia Neurons Is Modulated by Gut-Derived Mediators
Abstract
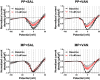
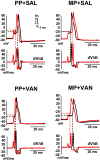
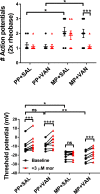
In the clinical setting, analgesic tolerance is a primary driver of diminished pain control and opioid dose escalations. Integral to this process are primary afferent sensory neurons, the first-order components of nociceptive sensation. Here, we characterize the factors modulating morphine action and tolerance in mouse small diameter dorsal root ganglia (DRG) neurons. We demonstrate that acute morphine inactivates tetrodotoxin-resistant (TTX-R) Na+ channels in these cells. Chronic exposure resulted in tolerance to this effect, which was prevented by treatment with oral vancomycin. Using colonic supernatants, we further show that mediators in the gut microenvironment of mice with chronic morphine exposure can induce tolerance and hyperexcitability in naive DRG neurons. Tolerance (but not hyperexcitability) in this paradigm was mitigated by oral vancomycin treatment. These findings collectively suggest that gastrointestinal microbiota modulate the development of morphine tolerance (but not hyperexcitability) in nociceptive primary afferent neurons, through a mechanism involving TTX-R Na+ channels.
Conflict of interest statement
DECLARATION OF INTERESTS All authors declare no conflict of interest or competing financial interests.
Figures








References
-
- Aghajanian G., Wang Y. Common alpha-2 and opiate effector mechanisms in the locus ceruleus: intracellular studies in brain slices. Neuropharmacology. 1987;26:789–800. - PubMed
-
- Akins P., McCleskey E. Characterization of potassium currents in adult rat sensory neurons and modulation by opioids and cyclic AMP. Neuroscience. 1993;56:759–769. - PubMed
-
- Amaya F., Decosterd I., Samad T.A., Plumpton C., Tate S., Mannion R.J., Costigan M., Woolf C.J. Diversity of expression of the sensory neuron-specific TTX-resistant voltage-gated sodium ion channels SNS and SNS2. Mol. Cell. Neurosci. 2000;15:331–342. - PubMed
-
- Angst M.S., Clark J.D. Opioid-induced hyperalgesia: a qualitative systematic review. Anesthesiology. 2006;104:570–587. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

