Roles of TRPV1 and TRPA1 in Spontaneous Pain from Inflamed Masseter Muscle
- PMID: 29890293
- PMCID: PMC6086573
- DOI: 10.1016/j.neuroscience.2018.05.048
Roles of TRPV1 and TRPA1 in Spontaneous Pain from Inflamed Masseter Muscle
Abstract
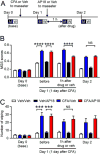
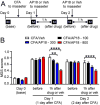
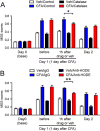
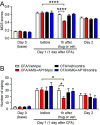
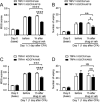
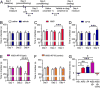
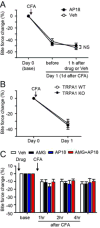
Craniofacial muscle pain, such as spontaneous pain and bite-evoked pain, are major symptoms in patients with temporomandibular disorders and infection. However, the underlying mechanisms of muscle pain, especially mechanisms of highly prevalent spontaneous pain, are poorly understood. Recently, we reported that transient receptor potential vanilloid 1 (TRPV1) contributes to spontaneous pain but only marginally contributes to bite-evoked pain during masseter inflammation. Here, we investigated the role of transient receptor potential ankyrin 1 (TRPA1) in spontaneous and bite-evoked pain during masseter inflammation, and dissected the relative contributions of TRPA1 and TRPV1. Masseter inflammation increased mouse grimace scale (MGS) scores and face wiping behaviors. Pharmacological or genetic inhibition of TRPA1 significantly attenuated MGS but not face wiping behaviors. MGS scores were also attenuated by scavenging putative endogenous ligands for TRPV1 or TRPA1. Simultaneous inhibition of TRPA1 by AP18 and TRPV1 by AMG9810 in masseter muscle resulted in robust inhibition of both MGS and face wiping behaviors. Administration of AP18 or AMG9810 to masseter muscle induced conditioned place preference (CPP). The extent of CPP following simultaneous administration of AP18 and AMG9810 was greater than that induced by the individual antagonists. In contrast, inflammation-induced reduction of bite force was not affected by the inhibition of TRPA1 alone or in combination with TRPV1. These results suggest that simultaneous inhibition of TRPV1 and TRPA1 produces additive relief of spontaneous pain, but does not ameliorate bite-evoked pain during masseter inflammation. Our results provide further evidence that distinct mechanisms underlie spontaneous and bite-evoked pain from inflamed masseter muscle.
Keywords: TRP channels; inflammation; muscle pain; trigeminal pain.
Copyright © 2018 IBRO. Published by Elsevier Ltd. All rights reserved.
Figures
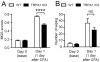
Similar articles
-
Is TRPA1 Burning Down TRPV1 as Druggable Target for the Treatment of Chronic Pain?Int J Mol Sci. 2019 Jun 14;20(12):2906. doi: 10.3390/ijms20122906. Int J Mol Sci. 2019. PMID: 31197115 Free PMC article. Review.
-
Spontaneous and Bite-Evoked Muscle Pain Are Mediated by a Common Nociceptive Pathway With Differential Contribution by TRPV1.J Pain. 2017 Nov;18(11):1333-1345. doi: 10.1016/j.jpain.2017.06.005. Epub 2017 Jun 29. J Pain. 2017. PMID: 28669862 Free PMC article.
-
TRPV1 and TRPA1 in Lung Inflammation and Airway Hyperresponsiveness Induced by Fine Particulate Matter (PM2.5).Oxid Med Cell Longev. 2019 Jun 2;2019:7450151. doi: 10.1155/2019/7450151. eCollection 2019. Oxid Med Cell Longev. 2019. PMID: 31281589 Free PMC article.
-
Activation of TRPV1 and TRPA1 leads to muscle nociception and mechanical hyperalgesia.Pain. 2009 Aug;144(3):270-277. doi: 10.1016/j.pain.2009.04.021. Epub 2009 May 22. Pain. 2009. PMID: 19464796 Free PMC article.
-
TRP channels in cancer pain.Eur J Pharmacol. 2021 Aug 5;904:174185. doi: 10.1016/j.ejphar.2021.174185. Epub 2021 May 17. Eur J Pharmacol. 2021. PMID: 34015320 Review.
Cited by
-
TRPV1 Channel Contributes to the Behavioral Hypersensitivity in a Rat Model of Complex Regional Pain Syndrome Type 1.Front Pharmacol. 2019 Apr 26;10:453. doi: 10.3389/fphar.2019.00453. eCollection 2019. Front Pharmacol. 2019. PMID: 31105572 Free PMC article.
-
Ablation of TRPV1+ Afferent Terminals by Capsaicin Mediates Long-Lasting Analgesia for Trigeminal Neuropathic Pain.eNeuro. 2020 May 29;7(3):ENEURO.0118-20.2020. doi: 10.1523/ENEURO.0118-20.2020. Print 2020 May/Jun. eNeuro. 2020. PMID: 32404326 Free PMC article.
-
TMEM100, a regulator of TRPV1-TRPA1 interaction, contributes to temporomandibular disorder pain.Front Mol Neurosci. 2023 Mar 23;16:1160206. doi: 10.3389/fnmol.2023.1160206. eCollection 2023. Front Mol Neurosci. 2023. PMID: 37033371 Free PMC article.
-
Is TRPA1 Burning Down TRPV1 as Druggable Target for the Treatment of Chronic Pain?Int J Mol Sci. 2019 Jun 14;20(12):2906. doi: 10.3390/ijms20122906. Int J Mol Sci. 2019. PMID: 31197115 Free PMC article. Review.
-
Biological action of bleaching agents on tooth structure: A review.Histol Histopathol. 2024 Oct;39(10):1229-1243. doi: 10.14670/HH-18-726. Epub 2024 Feb 28. Histol Histopathol. 2024. PMID: 38477344 Review.
References
-
- Castrillon EE, Cairns BE, Ernberg M, Wang K, Sessle BJ, Arendt-Nielsen L, Svensson P. Effect of peripheral NMDA receptor blockade with ketamine on chronic myofascial pain in temporomandibular disorder patients: a randomized, double-blinded, placebo-controlled trial. J Orofac Pain. 2008;22:122–130. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

