Phase I Study To Evaluate the Pharmacokinetics, Safety, and Tolerability of Two Dosing Regimens of Oral Fosfomycin Tromethamine in Healthy Adult Participants
- PMID: 29891606
- PMCID: PMC6105781
- DOI: 10.1128/AAC.00464-18
Phase I Study To Evaluate the Pharmacokinetics, Safety, and Tolerability of Two Dosing Regimens of Oral Fosfomycin Tromethamine in Healthy Adult Participants
Abstract
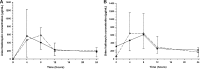
The pharmacokinetics (PK), safety, and tolerability of two repeated dosing regimens of oral fosfomycin tromethamine were evaluated in 18 healthy adult subjects. Subjects received 3 g every other day (QOD) for 3 doses and then every day (QD) for 7 doses, or vice versa, in a phase I, randomized, open-label, two-period-crossover study. Serial blood (n = 11) and urine (n = 4 collection intervals) samples were collected before and up to 24 h after dosing on days 1 and 5, along with predose concentrations on days 3 and 7. PK parameters were similar between days 1 and 5 within and between dosing regimens. The mean (± standard deviation [SD]) PK parameters for fosfomycin in plasma on day 5 during the respective QOD and QD dosing regimens were as follows: maximum concentration of drug in serum (Cmax) = 24.4 ± 6.2 versus 23.8 ± 5.6 μg/ml, time to Cmax (Tmax) = 2.2 ± 0.7 versus 2.0 ± 0.4 h, apparent volume of distribution (V/F) = 141 ± 67.9 versus 147 ± 67.6 liters, apparent clearance (CL/F) = 21.4 ± 8.0 versus 20.4 ± 5.3 liters/h, renal clearance (CLR) = 7.5 ± 4.1 versus 7.3 ± 3.5 liters/h, area under the concentration-time curve from 0 to 24 h (AUC0-24) = 151.6 ± 35.6 versus 156.6 ± 42.5 μg · h/ml, and elimination half-life (t1/2) = 4.5 ± 1.1 versus 5.0 ± 1.7 h. Urine concentrations peaked at approximately 600 μg/ml through the 0- to 8-h urine collection intervals but displayed significant interindividual variability. Roughly 35 to 40% of the 3-g dose was excreted in the urine by 24 h postdose. No new safety concerns were identified during this study. The proportion of diarrhea-free days during the study was significantly lower with the QD regimen than with the QOD regimen (61% versus 77%; P < 0.0001). Further studies to establish the clinical benefit/risk ratio for repeated dosing regimens of oral fosfomycin tromethamine are warranted. (This trial is registered at ClinicalTrials.gov under registration no. NCT02570074.).
Keywords: antimicrobial safety; fosfomycin; pharmacokinetics; safety; tolerability.
Copyright © 2018 American Society for Microbiology.
Figures



References
-
- Anonymous. July 2007. Monurol (fosfomycin tromethamine) package insert. Forest Pharmaceuticals, Inc, St Louis, MO: http://www.accessdata.fda.gov/drugsatfda_docs/label/2008/050717s005lbl.pdf Accessed 11 May 2015.
-
- Anonymous. 2015. Fosfomycin trometamol package insert. Mercury Pharmaceuticals Limited, London, United Kingdom: https://www.medicines.org.uk/emc/product/7219/smpc Accessed 30 May 2018.
-
- Gupta K, Hooton TM, Naber KG, Wullt B, Colgan R, Miller LG, Moran GJ, Nicolle LE, Raz R, Schaeffer AJ, Soper DE. 2011. International clinical practice guidelines for the treatment of acute uncomplicated cystitis and pyelonephritis in women: a 2010 update by the Infectious Diseases Society of America and the European Society for Microbiology and Infectious Diseases. Clin Infect Dis 52:e103–e120. doi: 10.1093/cid/ciq257. - DOI - PubMed
-
- Qiao LD, Zheng B, Chen S, Yang Y, Zhang K, Guo HF, Yang B, Niu YJ, Wang Y, Shi BK, Yang WM, Zhao XK, Gao XF, Chen M. 2013. Evaluation of three-dose fosfomycin tromethamine in the treatment of patients with urinary tract infections: an uncontrolled, open-label, multicentre study. BMJ Open 3:e004157. doi: 10.1136/bmjopen-2013-004157. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

