A Method Using Goldmann Stimulus Sizes I to V-Measured Sensitivities to Predict Lead Time Gained to Visual Field Defect Detection in Early Glaucoma
- PMID: 29892496
- PMCID: PMC5993363
- DOI: 10.1167/tvst.7.3.17
A Method Using Goldmann Stimulus Sizes I to V-Measured Sensitivities to Predict Lead Time Gained to Visual Field Defect Detection in Early Glaucoma
Abstract
Purpose: To predict the lead time (difference in time taken for a visual field [VF] defect to be detected) obtained when using stimulus sizes within or near the size of the critical area of spatial summation (Ac), and to test these predictions using sensitivity measurements from a cohort of glaucoma patients.
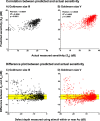
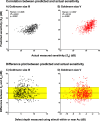
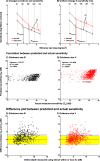
Methods: Thirty-seven patients with early open-angle glaucoma and 60 healthy observers underwent VF testing on the Humphrey Field Analyzer in full threshold mode using Goldmann stimulus sizes I to V (GI-V) across the 30-2 test grid. We used the sensitivities measured using GI to V in healthy patients to predict the lead time gained by using stimulus sizes within the size of Ac at all locations within the 30-2 grid. Then, we used sensitivities measured in the glaucoma patients to test this predictive model.
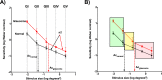
Results: Median lead time to VF defect detection when using stimulus sizes within Ac compared with stimulus sizes larger than Ac was 4.1 years across the 30-2 test grid (interquartile range, 3.1 and 5.1 years). Sensitivities of the glaucoma patients showed good agreement with the predictive model of lead time gained (77.5%-84.3% were within ±3 dB).
Conclusions: Our model predicted substantial lead time differences when using stimulus sizes within or near Ac. Such stimulus sizes could potentially detect VF defects, on average, 4 years earlier than current paradigms.
Translational relevance: Stimulus sizes within or near Ac may be more suitable for early detection of glaucomatous VF defects. Larger stimulus sizes may be more suitable for later monitoring of established disease.
Keywords: Humphrey Field Analyzer; Ricco's area; contrast sensitivity; perimetry; spatial summation.
Figures



References
-
- Jampel HD, Singh K, Lin SC, Chen TC, Francis BA, Hodapp E, Samples JR, Smith SD. . Assessment of visual function in glaucoma: a report by the American Academy of Ophthalmology. Ophthalmology. 2011; 118: 986– 1002. - PubMed
-
- Anderson RS. . The psychophysics of glaucoma: improving the structure/function relationship. Prog Retin Eye Res. 2006; 25: 79– 97. - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

