The P2X7 receptor and pannexin-1 are involved in glucose-induced autocrine regulation in β-cells
- PMID: 29895988
- PMCID: PMC5997690
- DOI: 10.1038/s41598-018-27281-9
The P2X7 receptor and pannexin-1 are involved in glucose-induced autocrine regulation in β-cells
Abstract
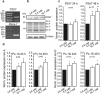
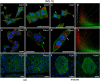
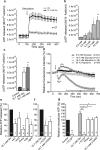
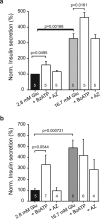
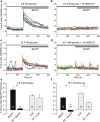
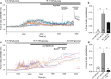
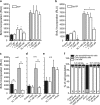
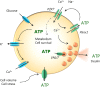
Extracellular ATP is an important short-range signaling molecule that promotes various physiological responses virtually in all cell types, including pancreatic β-cells. It is well documented that pancreatic β-cells release ATP through exocytosis of insulin granules upon glucose stimulation. We hypothesized that glucose might stimulate ATP release through other non-vesicular mechanisms. Several purinergic receptors are found in β-cells and there is increasing evidence that purinergic signaling regulates β-cell functions and survival. One of the receptors that may be relevant is the P2X7 receptor, but its detailed role in β-cell physiology is unclear. In this study we investigated roles of the P2X7 receptor and pannexin-1 in ATP release, intracellular ATP, Ca2+ signals, insulin release and cell proliferation/survival in β-cells. Results show that glucose induces rapid release of ATP and significant fraction of release involves the P2X7 receptor and pannexin-1, both expressed in INS-1E cells, rat and mouse β-cells. Furthermore, we provide pharmacological evidence that extracellular ATP, via P2X7 receptor, stimulates Ca2+ transients and cell proliferation in INS-1E cells and insulin secretion in INS-1E cells and rat islets. These data indicate that the P2X7 receptor and pannexin-1 have important functions in β-cell physiology, and should be considered in understanding and treatment of diabetes.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

