Tracking the sarcoplasmic reticulum membrane voltage in muscle with a FRET biosensor
- PMID: 29899059
- PMCID: PMC6080890
- DOI: 10.1085/jgp.201812035
Tracking the sarcoplasmic reticulum membrane voltage in muscle with a FRET biosensor
Abstract
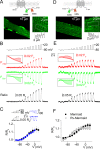
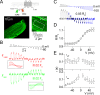
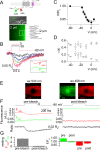
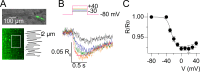
Ion channel activity in the plasma membrane of living cells generates voltage changes that are critical for numerous biological functions. The membrane of the endoplasmic/sarcoplasmic reticulum (ER/SR) is also endowed with ion channels, but whether changes in its voltage occur during cellular activity has remained ambiguous. This issue is critical for cell functions that depend on a Ca2+ flux across the reticulum membrane. This is the case for contraction of striated muscle, which is triggered by opening of ryanodine receptor Ca2+ release channels in the SR membrane in response to depolarization of the transverse invaginations of the plasma membrane (the t-tubules). Here, we use targeted expression of voltage-sensitive fluorescence resonance energy transfer (FRET) probes of the Mermaid family in differentiated muscle fibers to determine whether changes in SR membrane voltage occur during depolarization-contraction coupling. In the absence of an SR targeting sequence, FRET signals from probes present in the t-tubule membrane allow calibration of the voltage sensitivity and amplitude of the response to voltage-clamp pulses. Successful SR targeting of the probes was achieved using an N-terminal domain of triadin, which completely eliminates voltage-clamp-activated FRET signals from the t-tubule membrane of transfected fibers. In fibers expressing SR-targeted Mermaid probes, activation of SR Ca2+ release in the presence of intracellular ethyleneglycol-bis(β-amino-ethyl ether)-N,N,N',N'-tetra acetic acid (EGTA) results in an accompanying FRET signal. We find that this signal results from pH sensitivity of the probe, which detects cytosolic acidification because of the release of protons upon Ca2+ binding to EGTA. When EGTA is substituted with either 1,2-bis(o-aminophenoxy)ethane-N,N,N',N'-tetraacetic acid or the contraction blocker N-benzyl-p-toluene sulfonamide, we find no indication of a substantial change in the FRET response caused by a voltage change. These results suggest that the ryanodine receptor-mediated SR Ca2+ efflux is well balanced by concomitant counterion currents across the SR membrane.
© 2018 Sanchez et al.
Figures



Comment in
-
No voltage change at skeletal muscle SR membrane during Ca2+ release-just Mermaids on acid.J Gen Physiol. 2018 Aug 6;150(8):1055-1058. doi: 10.1085/jgp.201812084. Epub 2018 Jul 3. J Gen Physiol. 2018. PMID: 29970411 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

