CRISPR therapeutic tools for complex genetic disorders and cancer (Review)
- PMID: 29901119
- PMCID: PMC6017271
- DOI: 10.3892/ijo.2018.4434
CRISPR therapeutic tools for complex genetic disorders and cancer (Review)
Abstract
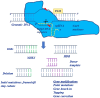
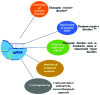
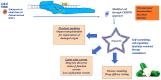
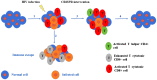
One of the fundamental discoveries in the field of biology is the ability to modulate the genome and to monitor the functional outputs derived from genomic alterations. In order to unravel new therapeutic options, scientists had initially focused on inducing genetic alterations in primary cells, in established cancer cell lines and mouse models using either RNA interference or cDNA overexpression or various programmable nucleases [zinc finger nucleases (ZNF), transcription activator-like effector nucleases (TALEN)]. Even though a huge volume of data was produced, its use was neither cheap nor accurate. Therefore, the clustered regularly interspaced short palindromic repeats (CRISPR) system was evidenced to be the next step in genome engineering tools. CRISPR-associated protein 9 (Cas9)-mediated genetic perturbation is simple, precise and highly efficient, empowering researchers to apply this method to immortalized cancerous cell lines, primary cells derived from mouse and human origins, xenografts, induced pluripotent stem cells, organoid cultures, as well as the generation of genetically engineered animal models. In this review, we assess the development of the CRISPR system and its therapeutic applications to a wide range of complex diseases (particularly distinct tumors), aiming at personalized therapy. Special emphasis is given to organoids and CRISPR screens in the design of innovative therapeutic approaches. Overall, the CRISPR system is regarded as an eminent genome engineering tool in therapeutics. We envision a new era in cancer biology during which the CRISPR-based genome engineering toolbox will serve as the fundamental conduit between the bench and the bedside; nonetheless, certain obstacles need to be addressed, such as the eradication of side-effects, maximization of efficiency, the assurance of delivery and the elimination of immunogenicity.
Figures

References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

