Exposure to heavy metal stress triggers changes in plasmodesmatal permeability via deposition and breakdown of callose
- PMID: 29901781
- PMCID: PMC6022669
- DOI: 10.1093/jxb/ery171
Exposure to heavy metal stress triggers changes in plasmodesmatal permeability via deposition and breakdown of callose
Abstract
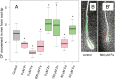
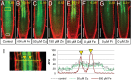
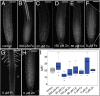
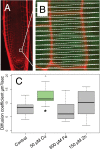
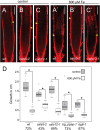
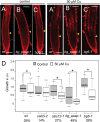
Both plants and animals must contend with changes in their environment. The ability to respond appropriately to these changes often underlies the ability of the individual to survive. In plants, an early response to environmental stress is an alteration in plasmodesmatal permeability with accompanying changes in cell to cell signaling. However, the ways in which plasmodesmata are modified, the molecular players involved in this regulation, and the biological significance of these responses are not well understood. Here, we examine the effects of nutrient scarcity and excess on plasmodesmata-mediated transport in the Arabidopsis thaliana root and identify two CALLOSE SYNTHASES and two β-1,3-GLUCANASES as key regulators of these processes. Our results suggest that modification of plasmodesmata-mediated signaling underlies the ability of the plant to maintain root growth and properly partition nutrients when grown under conditions of excess nutrients.
Figures



Similar articles
-
Heat Stress Reduces Root Meristem Size via Induction of Plasmodesmal Callose Accumulation Inhibiting Phloem Unloading in Arabidopsis.Int J Mol Sci. 2022 Feb 13;23(4):2063. doi: 10.3390/ijms23042063. Int J Mol Sci. 2022. PMID: 35216183 Free PMC article.
-
Analysis of a novel mutant allele of GSL8 reveals its key roles in cytokinesis and symplastic trafficking in Arabidopsis.BMC Plant Biol. 2018 Nov 22;18(1):295. doi: 10.1186/s12870-018-1515-y. BMC Plant Biol. 2018. PMID: 30466394 Free PMC article.
-
Arabidopsis callose synthases CalS1/8 regulate plasmodesmal permeability during stress.Nat Plants. 2016 Apr 11;2(5):16034. doi: 10.1038/nplants.2016.34. Nat Plants. 2016. PMID: 27243643
-
Biology of callose (β-1,3-glucan) turnover at plasmodesmata.Protoplasma. 2011 Jan;248(1):117-30. doi: 10.1007/s00709-010-0247-0. Epub 2010 Nov 30. Protoplasma. 2011. PMID: 21116665 Free PMC article. Review.
-
Callose balancing at plasmodesmata.J Exp Bot. 2018 Nov 26;69(22):5325-5339. doi: 10.1093/jxb/ery317. J Exp Bot. 2018. PMID: 30165704 Review.
Cited by
-
Advances in understanding the graft healing mechanism: a review of factors and regulatory pathways.Hortic Res. 2024 Jun 20;11(8):uhae175. doi: 10.1093/hr/uhae175. eCollection 2024 Aug. Hortic Res. 2024. PMID: 39108577 Free PMC article.
-
Alleviation of cadmium toxicity in Zea mays L. through up-regulation of growth, antioxidant defense system and organic osmolytes under calcium supplementation.PLoS One. 2022 Jun 22;17(6):e0269162. doi: 10.1371/journal.pone.0269162. eCollection 2022. PLoS One. 2022. PMID: 35731737 Free PMC article.
-
Iron availability modulates the Arabidopsis thaliana root calcium signature evoked by exogenous ATP.Plant Signal Behav. 2019;14(9):1640563. doi: 10.1080/15592324.2019.1640563. Epub 2019 Jul 13. Plant Signal Behav. 2019. PMID: 31304865 Free PMC article.
-
Comparative proteomic study of phytotoxic effects of silver nanoparticles and silver ions on tobacco plants.Environ Sci Pollut Res Int. 2019 Aug;26(22):22529-22550. doi: 10.1007/s11356-019-05552-w. Epub 2019 Jun 3. Environ Sci Pollut Res Int. 2019. PMID: 31161543
-
Lead transfer in the soil-root-plant system in a highly contaminated Andean area.PeerJ. 2021 Jan 5;9:e10624. doi: 10.7717/peerj.10624. eCollection 2021. PeerJ. 2021. PMID: 33505801 Free PMC article.
References
-
- Anderson MA. 1991. Long-term effects of copper rich swine manure application on continuous corn production. Communications in Soil Science and Plant Analysis 22, 993–1002.
-
- Angell SM, Davies C, Baulcombe DC. 1996. Cell-to-cell movement of potato virus X is associated with a change in the size-exclusion limit of plasmodesmata in trichome cells of Nicotiana clevelandii. Virology 216, 197–201. - PubMed
-
- Arias M, López E, Fernández D, Soto B. 2004. Copper distribution and dynamics in acid vineyard soils treated with copper-based fungicides. Soil Science 169, 796–805.
-
- Bates TE. 1971. Factors affecting critical nutrient concentrations in plants and their evaluation: a review. Soil Science 112, 116–130.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

