Engineering cardiac microphysiological systems to model pathological extracellular matrix remodeling
- PMID: 29906229
- PMCID: PMC6230901
- DOI: 10.1152/ajpheart.00110.2018
Engineering cardiac microphysiological systems to model pathological extracellular matrix remodeling
Abstract
Many cardiovascular diseases are associated with pathological remodeling of the extracellular matrix (ECM) in the myocardium. ECM remodeling is a complex, multifactorial process that often contributes to declines in myocardial function and progression toward heart failure. However, the direct effects of the many forms of ECM remodeling on myocardial cell and tissue function remain elusive, in part because conventional model systems used to investigate these relationships lack robust experimental control over the ECM. To address these shortcomings, microphysiological systems are now being developed and implemented to establish direct relationships between distinct features in the ECM and myocardial function with unprecedented control and resolution in vitro. In this review, we will first highlight the most prominent characteristics of ECM remodeling in cardiovascular disease and describe how these features can be mimicked with synthetic and natural biomaterials that offer independent control over multiple ECM-related parameters, such as rigidity and composition. We will then detail innovative microfabrication techniques that enable precise regulation of cellular architecture in two and three dimensions. We will also describe new approaches for quantifying multiple aspects of myocardial function in vitro, such as contractility, action potential propagation, and metabolism. Together, these collective technologies implemented as cardiac microphysiological systems will continue to uncover important relationships between pathological ECM remodeling and myocardial cell and tissue function, leading to new fundamental insights into cardiovascular disease, improved human disease models, and novel therapeutic approaches.
Keywords: biomaterials; contractility; electrophysiology; metabolism; microfabrication.
Figures

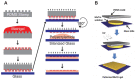
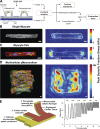
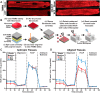

Similar articles
-
Inflammation-mediated matrix remodeling of extracellular matrix-mimicking biomaterials in tissue engineering and regenerative medicine.Acta Biomater. 2022 Oct 1;151:106-117. doi: 10.1016/j.actbio.2022.08.015. Epub 2022 Aug 13. Acta Biomater. 2022. PMID: 35970482 Review.
-
Biohybrid cardiac ECM-based hydrogels improve long term cardiac function post myocardial infarction.Acta Biomater. 2017 Mar 1;50:220-233. doi: 10.1016/j.actbio.2016.12.015. Epub 2016 Dec 9. Acta Biomater. 2017. PMID: 27956366
-
Applications of Cardiac Extracellular Matrix in Tissue Engineering and Regenerative Medicine.Adv Exp Med Biol. 2018;1098:59-83. doi: 10.1007/978-3-319-97421-7_4. Adv Exp Med Biol. 2018. PMID: 30238366 Review.
-
Hybrid and Composite Scaffolds Based on Extracellular Matrices for Cartilage Tissue Engineering.Tissue Eng Part B Rev. 2019 Jun;25(3):202-224. doi: 10.1089/ten.TEB.2018.0245. Tissue Eng Part B Rev. 2019. PMID: 30648478 Review.
-
Role of Extracellular Matrix in Cardiac Cellular Therapies.Adv Exp Med Biol. 2018;1098:173-188. doi: 10.1007/978-3-319-97421-7_9. Adv Exp Med Biol. 2018. PMID: 30238371 Review.
Cited by
-
Bioengineering approaches to mature induced pluripotent stem cell-derived atrial cardiomyocytes to model atrial fibrillation.Exp Biol Med (Maywood). 2021 Aug;246(16):1816-1828. doi: 10.1177/15353702211009146. Epub 2021 Apr 25. Exp Biol Med (Maywood). 2021. PMID: 33899540 Free PMC article. Review.
-
An American Physiological Society cross-journal Call for Papers on "Deconstructing Organs: Single-Cell Analyses, Decellularized Organs, Organoids, and Organ-on-a-Chip Models".Am J Physiol Lung Cell Mol Physiol. 2020 Aug 1;319(2):L266-L272. doi: 10.1152/ajplung.00311.2020. Epub 2020 Jul 1. Am J Physiol Lung Cell Mol Physiol. 2020. PMID: 32609556 Free PMC article. No abstract available.
-
3D-bioprinted cardiac tissues and their potential for disease modeling.J 3D Print Med. 2023 Jun;7(2):10.2217/3dp-2022-0023. doi: 10.2217/3dp-2022-0023. Epub 2023 Apr 4. J 3D Print Med. 2023. PMID: 38250545 Free PMC article.
-
Engineering the Cellular Microenvironment of Post-infarct Myocardium on a Chip.Front Cardiovasc Med. 2021 Jul 14;8:709871. doi: 10.3389/fcvm.2021.709871. eCollection 2021. Front Cardiovasc Med. 2021. PMID: 34336962 Free PMC article. Review.
-
Biomimetic Approaches in Cardiac Tissue Engineering: Replicating the Native Heart Microenvironment.Cureus. 2023 Aug 13;15(8):e43431. doi: 10.7759/cureus.43431. eCollection 2023 Aug. Cureus. 2023. PMID: 37581196 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources

