Evolution of New Delhi metallo-β-lactamase (NDM) in the clinic: Effects of NDM mutations on stability, zinc affinity, and mono-zinc activity
- PMID: 29909397
- PMCID: PMC6093243
- DOI: 10.1074/jbc.RA118.003835
Evolution of New Delhi metallo-β-lactamase (NDM) in the clinic: Effects of NDM mutations on stability, zinc affinity, and mono-zinc activity
Abstract
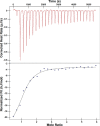
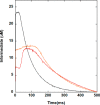
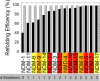
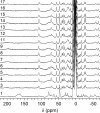
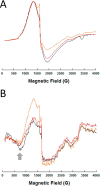
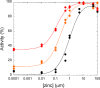
Infections by carbapenem-resistant Enterobacteriaceae are difficult to manage owing to broad antibiotic resistance profiles and because of the inability of clinically used β-lactamase inhibitors to counter the activity of metallo-β-lactamases often harbored by these pathogens. Of particular importance is New Delhi metallo-β-lactamase (NDM), which requires a di-nuclear zinc ion cluster for catalytic activity. Here, we compare the structures and functions of clinical NDM variants 1-17. The impact of NDM variants on structure is probed by comparing melting temperature and refolding efficiency and also by spectroscopy (UV-visible, 1H NMR, and EPR) of di-cobalt metalloforms. The impact of NDM variants on function is probed by determining the minimum inhibitory concentrations of various antibiotics, pre-steady-state and steady-state kinetics, inhibitor binding, and zinc dependence of resistance and activity. We observed only minor differences among the fully loaded di-zinc enzymes, but most NDM variants had more distinguishable selective advantages in experiments that mimicked zinc scarcity imposed by typical host defenses. Most NDM variants exhibited improved thermostability (up to ∼10 °C increased Tm ) and improved zinc affinity (up to ∼10-fold decreased Kd, Zn2). We also provide first evidence that some NDM variants have evolved the ability to function as mono-zinc enzymes with high catalytic efficiency (NDM-15, ampicillin: kcat/Km = 5 × 106 m-1 s-1). These findings reveal the molecular mechanisms that NDM variants have evolved to overcome the combined selective pressures of β-lactam antibiotics and zinc deprivation.
Keywords: Co(II)-substituted enzyme; NDM-1; antibiotic resistance; antibiotics; enzyme kinetics; enzyme mutation; metallo-β-lactamase; metalloenzyme; protein evolution.
Conflict of interest statement
The authors declare that they have no conflicts of interest with the contents of this article
Figures



References
-
- Solomon S. L., and Oliver K. B. (2014) Antibiotic resistance threats in the United States: stepping back from the brink. Am. Fam. Physician 89, 938–941 - PubMed
-
- Woodworth K. R., Walters M. S., Weiner L. M., Edwards J., Brown A. C., Huang J. Y., Malik S., Slayton R. B., Paul P., Capers C., Kainer M. A., Wilde N., Shugart A., Mahon G., Kallen A. J., Patel J., McDonald L. C., Srinivasan A., Craig M., and Cardo D. M. (2018) Vital signs: containment of novel multidrug-resistant organisms and resistance mechanisms–United States, 2006–2017. MMWR Morb. Mortal. Wkly Rep. 67, 396–401 10.15585/mmwr.mm6713e1 - DOI - PMC - PubMed
-
- Makena A., Brem J., Pfeffer I., Geffen R. E., Wilkins S. E., Tarhonskaya H., Flashman E., Phee L. M., Wareham D. W., and Schofield C. J. (2015) Biochemical characterization of New Delhi metallo-β-lactamase variants reveals differences in protein stability. J. Antimicrob. Chemother. 70, 463–469 10.1093/jac/dku403 - DOI - PMC - PubMed
-
- Stewart A. C., Bethel C. R., VanPelt J., Bergstrom A., Cheng Z., Miller C. G., Williams C., Poth R., Morris M., Lahey O., Nix J. C., Tierney D. L., Page R. C., Crowder M. W., Bonomo R. A., and Fast W. (2017) Clinical variants of New Delhi metallo-β-lactamase are evolving to overcome zinc scarcity. ACS Infect. Dis. 3, 927–940 10.1021/acsinfecdis.7b00128 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

