Performance status dynamics during treatment with nab-paclitaxel plus gemcitabine versus gemcitabine alone for metastatic pancreatic cancer
- PMID: 29910636
- PMCID: PMC5987855
- DOI: 10.2147/CMAR.S163475
Performance status dynamics during treatment with nab-paclitaxel plus gemcitabine versus gemcitabine alone for metastatic pancreatic cancer
Abstract
Objectives: This analysis examined changes in Karnofsky performance status (KPS) as a surrogate for patient's well-being during treatment with nab-paclitaxel plus gemcitabine vs gemcitabine alone as first-line therapy for metastatic pancreatic cancer (MPC) in the Phase III MPACT trial.
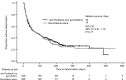
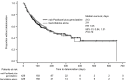
Participants and methods: Descriptive analyses were performed for KPS at three time points (3 and 6 months after randomization and 1 month before disease progression) and for time to any KPS deterioration. Time to definitive KPS deterioration (≥10-point KPS decrease from baseline) was calculated using the Kaplan-Meier method. A larger decrease from baseline (≥20 points) was investigated as a sensitivity analysis. A Cox proportional hazards model analyzed the effect of baseline factors (including treatment) potentially associated with time to definitive deterioration.
Results: The two treatment arms had generally comparable time to any KPS deterioration, similar KPS at 3 and 6 months after randomization and at 1 month before disease progression, and no significant difference in time to definitive deterioration. Baseline KPS, neutrophil-to-lymphocyte ratio, age, liver metastases, and region had a significant effect on time to definitive KPS deterioration, but treatment arm did not.
Conclusion: The increased survival observed with nab-paclitaxel plus gemcitabine was not associated with adverse effects on performance status.
Keywords: Karnofsky performance status; chemotherapy; gemcitabine; metastatic pancreatic cancer; nab-paclitaxel.
Conflict of interest statement
Disclosure EGC has received research funding from and participated in an advisory board for Celgene Corporation. DVH has served as a consultant or advisor for and received honoraria from Celgene Corporation and received research funding from HonorHealth. YW was an employee of Pharmerit International, which received research funding from Celgene Corporation. SM-D is an employee of Celgene Corporation. MB is an employee and shareholder of Pharmerit International, which received research funding from Celgene Corporation. DG has received research funding from Celgene Corporation and Pfizer and served as a consultant or advisor (unremunerated) for Celgene Corporation and Pfizer. The authors report no other conflicts of interest in this work.
Figures

References
-
- SEER Stat Facts Sheets: Pancreas Cancer. 2017. [Accessed April 17, 2018]. [webpage on the Internet] Available from: http://seer.cancer.gov/statfacts/html/pancreas.html.
-
- Clark KL, Loscalzo M, Trask PC, Zabora J, Philip EJ. Psychological distress in patients with pancreatic cancer – an understudied group. Psychooncology. 2010;19(12):1313–1320. - PubMed
-
- Engebretson A, Matrisian L, Thompson C. Pancreatic cancer: patient and caregiver perceptions on diagnosis, psychological impact, and importance of support. Pancreatology. 2015;15(6):701–707. - PubMed
-
- Kristensen A, Vagnildhaug OM, Grønberg BH, Kaasa S, Laird B, Solheim TS. Does chemotherapy improve health-related quality of life in advanced pancreatic cancer? A systematic review. Crit Rev Oncol Hematol. 2016;99:286–298. - PubMed
-
- National Comprehensive Cancer Network [Accessed April 17, 2018];NCCN Clinical Practice Guidelines in Oncology Pancreatic Adenocarcinoma. 2017 3 Available from: https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf.
LinkOut - more resources
Full Text Sources
Other Literature Sources

