Effect of two cosmetic compounds on the growth, biofilm formation activity, and surface properties of acneic strains of Cutibacterium acnes and Staphylococcus aureus
- PMID: 29911330
- PMCID: PMC6436439
- DOI: 10.1002/mbo3.659
Effect of two cosmetic compounds on the growth, biofilm formation activity, and surface properties of acneic strains of Cutibacterium acnes and Staphylococcus aureus
Abstract
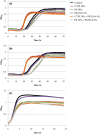
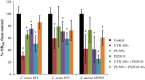
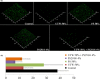
Increasing popularity of preservative-free cosmetics necessitates in-depth research, specifically as bacteria can react to local factors by important metabolic changes. In this respect, investigating the effect of cosmetic preparations on pathogenic strains of commensal species such as acneic forms of Cutibacterium acnes (former Propionibacterium acnes) and bacteria behaving both as commensals and opportunistic pathogens such as Staphylococcus aureus is of major interest. In this study, we studied the effect of commonly used cosmetics, Uriage™ thermal water (UTW) and a rhamnose-rich polysaccharide (PS291® ) on RT4 and RT5 acneic strains of C. acnes and a cutaneous strain of S. aureus. UTW affected the growth kinetic of acneic C. acnes essentially by increasing its generation time and reducing its biomass, whereas only the S. aureus final biomass was decreased. PS291 had more marginal effects. Both compounds showed a marked antibiofilm activity on C. acnes and S. aureus. For S. aureus that appeared essentially due to inhibition of initial adhesion. Cosmetics did not modify the metabolic activity of bacteria. Both C. acnes and S. aureus showed marked hydrophobic surface properties. UTW and PS291 had limited effect on C. acnes but increased the hydrophobic character of S. aureus. This work underlines the effect of cosmetics on cutaneous bacteria and the potential limitations of preservative-free products.
Keywords: Cutibacterium acnes; Staphylococcus aureus; biofilm; cosmetics; metabolism; surface adhesion; surface polarity.
© 2018 The Authors. MicrobiologyOpen published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors have the following interests. This work was supported by public funds obtained from Evreux Porte de Normandie, Region Normandie and European Union (FEDER). Luc Lefeuvre is employed by the Dermatologic Laboratories Uriage. There are no patents, products in development of marketed products to declare. This does not alter the authors’ adherence to all policies on sharing data and materials, as detailed online in the guide for authors.
Figures



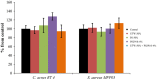
Similar articles
-
Effects of Ulva sp. Extracts on the Growth, Biofilm Production, and Virulence of Skin Bacteria Microbiota: Staphylococcus aureus, Staphylococcus epidermidis, and Cutibacterium acnes Strains.Molecules. 2021 Aug 6;26(16):4763. doi: 10.3390/molecules26164763. Molecules. 2021. PMID: 34443349 Free PMC article.
-
Adaptation of acneic and non acneic strains of Cutibacterium acnes to sebum-like environment.Microbiologyopen. 2019 Sep;8(9):e00841. doi: 10.1002/mbo3.841. Epub 2019 Apr 4. Microbiologyopen. 2019. PMID: 30950214 Free PMC article.
-
Antibiofilm activities of fatty acids including myristoleic acid against Cutibacterium acnes via reduced cell hydrophobicity.Phytomedicine. 2021 Oct;91:153710. doi: 10.1016/j.phymed.2021.153710. Epub 2021 Aug 18. Phytomedicine. 2021. PMID: 34461422
-
Recent advances in understanding Propionibacterium acnes ( Cutibacterium acnes) in acne.F1000Res. 2018 Dec 19;7:F1000 Faculty Rev-1953. doi: 10.12688/f1000research.15659.1. eCollection 2018. F1000Res. 2018. PMID: 30613388 Free PMC article. Review.
-
Staphylococcus epidermidis and Cutibacterium acnes: Two Major Sentinels of Skin Microbiota and the Influence of Cosmetics.Microorganisms. 2020 Nov 7;8(11):1752. doi: 10.3390/microorganisms8111752. Microorganisms. 2020. PMID: 33171837 Free PMC article. Review.
Cited by
-
Microbiome and Probiotics in Acne Vulgaris-A Narrative Review.Life (Basel). 2022 Mar 15;12(3):422. doi: 10.3390/life12030422. Life (Basel). 2022. PMID: 35330173 Free PMC article. Review.
-
Comprehensive review of dermatological and cosmeceutical manifestations of thermal water and future insights.Int J Biometeorol. 2025 Aug;69(8):1783-1817. doi: 10.1007/s00484-025-02937-0. Epub 2025 May 27. Int J Biometeorol. 2025. PMID: 40423748 Free PMC article. Review.
-
Skin Microbiome, Metabolome and Skin Phenome, from the Perspectives of Skin as an Ecosystem.Phenomics. 2022 Oct 10;2(6):363-382. doi: 10.1007/s43657-022-00073-y. eCollection 2022 Dec. Phenomics. 2022. PMID: 36939800 Free PMC article. Review.
-
Cotton and Flax Textiles Leachables Impact Differently Cutaneous Staphylococcus aureus and Staphylococcus epidermidis Biofilm Formation and Cytotoxicity.Life (Basel). 2022 Apr 6;12(4):535. doi: 10.3390/life12040535. Life (Basel). 2022. PMID: 35455029 Free PMC article.
-
Effects of Ulva sp. Extracts on the Growth, Biofilm Production, and Virulence of Skin Bacteria Microbiota: Staphylococcus aureus, Staphylococcus epidermidis, and Cutibacterium acnes Strains.Molecules. 2021 Aug 6;26(16):4763. doi: 10.3390/molecules26164763. Molecules. 2021. PMID: 34443349 Free PMC article.
References
-
- Aryee, A. , & Edgeworth, J. D. (2017). Carriage, clinical microbiology and transmission of Staphylococcus aureus . Current Topics in Microbiology and Immunology, 409, 1–19. - PubMed
-
- Bellon‐Fontaine, M. , Rault, J. , & van Oss, C. (1996). Microbial adhesion to solvents: A novel method to determine the electron‐donor/electron‐acceptor or Lewis acid‐base properties of microbial cells. Colloids and Surfaces B: Biointerfaces, 7, 47–53. 10.1016/0927-7765(96)01272-6 - DOI
-
- Berridge, M. , & Tan, A. (1993). Characterization of the cellular reduction of 3‐(4,5‐dimethylthiazol‐2‐yl)‐2,5‐diphenyltetrazolium bromide (MTT): Subcellular localization, substrate dependence, and involvement of mitochondrial electron transport in MTT reduction. Archives of Biochemistry and Biophysics, 303, 474–482. 10.1006/abbi.1993.1311 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

