A Randomized Controlled Trial of Balloon Dilation as a Treatment for Persistent Eustachian Tube Dysfunction With 1-Year Follow-Up
- PMID: 29912819
- PMCID: PMC6075883
- DOI: 10.1097/MAO.0000000000001853
A Randomized Controlled Trial of Balloon Dilation as a Treatment for Persistent Eustachian Tube Dysfunction With 1-Year Follow-Up
Abstract
Objective: Compare Eustachian tube balloon dilation versus continued medical therapy (control) for treating persistent Eustachian tube dysfunction (ETD).
Study design: Prospective, multicenter, randomized controlled trial.
Setting: Tertiary care academic center and private practice.
Patients: Diagnosed with medically refractory persistent ETD.
Interventions: 1:1 Randomization to balloon dilation or control. After 6 weeks, control participants had the option to undergo balloon dilation if symptoms persisted.
Main outcome measures: Primary efficacy endpoint was the comparison between treatment arms in the mean change from baseline in the 7-item Eustachian Tube Dysfunction Questionnaire (ETDQ-7) score. Primary safety endpoint was complication rate.
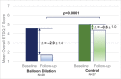
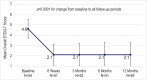
Results: Sixty participants were randomized (31 balloon dilation, 29 control). Mean (SD) change in overall ETDQ-7 score at 6 weeks was -2.9 (1.4) for balloon dilation compared with -0.6 (1.0) for control: balloon dilation was superior to control (p < 0.0001). No complications were reported in either study arm. Among participants with abnormal baseline assessments, improvements in tympanogram type (p < 0.006) and tympanic membrane position (p < 0.001) were significantly better for balloon dilation than control. Technical success was 100% (91 successful dilations/91 attempts) and most procedures (72%) were completed in the office under local anesthesia. Improvements in the ETDQ-7 scores were maintained through 12 months after balloon dilation.
Conclusions: Balloon dilation is a safe and effective treatment for persistent ETD. Based on improved ETDQ-7 scores, balloon dilation is superior to continued medical management for persistent ETD. Symptom improvement is durable through a minimum of 12 months. Procedures are well tolerated in the office setting under local anesthesia.
Figures
References
-
- Poe DS, Hanna BMN. Balloon dilation of the cartilaginous portion of the Eustachian tube: initial safety and feasibility analysis in a cadaver model. Am J Otolaryngol 2011; 32:115–123. - PubMed
-
- Ockermann T, Reineke U, Upile T, Ebmeyer J, Sudhoff HH. Balloon dilation Eustachian tuboplasty: a feasibility study. Otol Neurotol 2010; 31:1100–1103. - PubMed
-
- McCoul ED, Singh A, Anand VK, Tabaee A. Balloon dilation of the eustachian tube in a cadaver model: technical considerations, learning curve, and potential barriers. Laryngoscope 2012; 122:718–723. - PubMed
-
- Ockermann T, Reineke U, Upile T, Ebmeyer J, Sudhoff H. Balloon dilatation Eustachian tuboplasty: a clinical study. Laryngoscope 2010; 120:1411–1416. - PubMed
-
- Poe DS, Silvola J, Pyykko I. Balloon dilation of the cartilaginous Eustachian tube. Otolaryngol Head Neck Surg 2011; 144:563–569. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

