Long noncoding RNA repertoire and targeting by nuclear exosome, cytoplasmic exonuclease, and RNAi in fission yeast
- PMID: 29914874
- PMCID: PMC6097657
- DOI: 10.1261/rna.065524.118
Long noncoding RNA repertoire and targeting by nuclear exosome, cytoplasmic exonuclease, and RNAi in fission yeast
Abstract
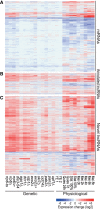
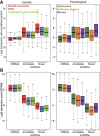
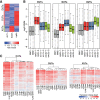
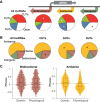
Long noncoding RNAs (lncRNAs), which are longer than 200 nucleotides but often unstable, contribute a substantial and diverse portion to pervasive noncoding transcriptomes. Most lncRNAs are poorly annotated and understood, although several play important roles in gene regulation and diseases. Here we systematically uncover and analyze lncRNAs in Schizosaccharomyces pombe. Based on RNA-seq data from twelve RNA-processing mutants and nine physiological conditions, we identify 5775 novel lncRNAs, nearly 4× the previously annotated lncRNAs. The expression of most lncRNAs becomes strongly induced under the genetic and physiological perturbations, most notably during late meiosis. Most lncRNAs are cryptic and suppressed by three RNA-processing pathways: the nuclear exosome, cytoplasmic exonuclease, and RNAi. Double-mutant analyses reveal substantial coordination and redundancy among these pathways. We classify lncRNAs by their dominant pathway into cryptic unstable transcripts (CUTs), Xrn1-sensitive unstable transcripts (XUTs), and Dicer-sensitive unstable transcripts (DUTs). XUTs and DUTs are enriched for antisense lncRNAs, while CUTs are often bidirectional and actively translated. The cytoplasmic exonuclease, along with RNAi, dampens the expression of thousands of lncRNAs and mRNAs that become induced during meiosis. Antisense lncRNA expression mostly negatively correlates with sense mRNA expression in the physiological, but not the genetic conditions. Intergenic and bidirectional lncRNAs emerge from nucleosome-depleted regions, upstream of positioned nucleosomes. Our results highlight both similarities and differences to lncRNA regulation in budding yeast. This broad survey of the lncRNA repertoire and characteristics in S. pombe, and the interwoven regulatory pathways that target lncRNAs, provides a rich framework for their further functional analyses.
Keywords: NMD pathway; RNA degradation; Schizosaccharomyces pombe; antisense RNA; cytoplasmic exonuclease; pervasive transcription.
© 2018 Atkinson et al.; Published by Cold Spring Harbor Laboratory Press for the RNA Society.
Figures


References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
