SNAI2 upregulation is associated with an aggressive phenotype in fulvestrant-resistant breast cancer cells and is an indicator of poor response to endocrine therapy in estrogen receptor-positive metastatic breast cancer
- PMID: 29921289
- PMCID: PMC6009053
- DOI: 10.1186/s13058-018-0988-9
SNAI2 upregulation is associated with an aggressive phenotype in fulvestrant-resistant breast cancer cells and is an indicator of poor response to endocrine therapy in estrogen receptor-positive metastatic breast cancer
Abstract
Background: Endocrine resistance in estrogen receptor-positive (ER+) breast cancer is a major clinical problem and is associated with accelerated cancer cell growth, increased motility and acquisition of mesenchymal characteristics. However, the specific molecules and pathways involved in these altered features remain to be detailed, and may be promising therapeutic targets to overcome endocrine resistance.
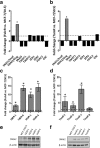
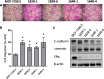
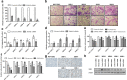
Methods: In the present study, we evaluated altered expression of epithelial-mesenchymal transition (EMT) regulators in ER+ breast cancer cell models of tamoxifen or fulvestrant resistance, by gene expression profiling. We investigated the specific role of increased SNAI2 expression in fulvestrant-resistant cells by gene knockdown and treatment with a SNAIL-p53 binding inhibitor, and evaluated the effect on cell growth, migration and expression of EMT markers. Furthermore, we evaluated SNAI2 expression by immunohistochemical analysis in metastatic samples from two cohorts of patients with breast cancer treated with endocrine therapy in the advanced setting.
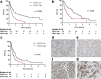
Results: SNAI2 was found to be significantly upregulated in all endocrine-resistant cells compared to parental cell lines, while no changes were observed in the expression of other EMT-associated transcription factors. SNAI2 knockdown with specific small interfering RNA (siRNA) converted the mesenchymal-like fulvestrant-resistant cells into an epithelial-like phenotype and reduced cell motility. Furthermore, inhibition of SNAI2 with specific siRNA or a SNAIL-p53 binding inhibitor reduced growth of cells resistant to fulvestrant treatment. Clinical evaluation of SNAI2 expression in two independent cohorts of patients with ER+ metastatic breast cancer treated with endocrine therapy in the advanced setting (N = 86 and N = 67) showed that high SNAI2 expression in the metastasis correlated significantly with shorter progression-free survival on endocrine treatment (p = 0.0003 and p = 0.004).
Conclusions: Our results suggest that SNAI2 is a key regulator of the aggressive phenotype observed in endocrine-resistant breast cancer cells, an independent prognostic biomarker in ER+ advanced breast cancer treated with endocrine therapy, and may be a promising therapeutic target in combination with endocrine therapies in ER+ metastatic breast cancer exhibiting high SNAI2 levels.
Keywords: Endocrine resistance; Epithelial-mesenchymal transition; Estrogen receptor-positive breast cancer; Fulvestrant; SNAI2.
Conflict of interest statement
Ethics approval and consent to participate
All clinical samples were coded to maintain patient confidentiality and studies were approved by the Ethics Committee of the Region of Southern Denmark (approval no S-2008-0115) and the Danish Data Protection Agency (approval no. 2008–580035(14/10607)).
Competing interests
The authors declare that they have no competing interests.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

