Caspase-3 Is a Pivotal Regulator of Microvascular Rarefaction and Renal Fibrosis after Ischemia-Reperfusion Injury
- PMID: 29925521
- PMCID: PMC6050936
- DOI: 10.1681/ASN.2017050581
Caspase-3 Is a Pivotal Regulator of Microvascular Rarefaction and Renal Fibrosis after Ischemia-Reperfusion Injury
Abstract
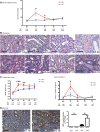
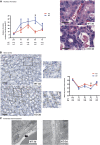
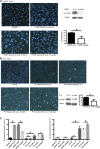
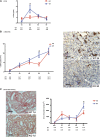
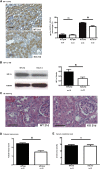
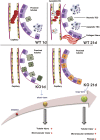
Background Ischemia-reperfusion injury (IRI) is a major risk factor for chronic renal failure. Here, we characterize the different modes of programmed cell death in the tubular and microvascular compartments during the various stages of IRI-induced AKI, and their relative importance to renal fibrogenesis.Methods We performed unilateral renal artery clamping for 30 minutes and contralateral nephrectomy in wild-type mice (C57BL/6) or caspase-3-/- mice.Results Compared with their wild-type counterparts, caspase-3-/- mice in the early stage of AKI had high urine cystatin C levels, tubular injury scores, and serum creatinine levels. Electron microscopy revealed evidence of tubular epithelial cell necrosis in caspase-3-/- mice, and immunohistochemistry showed upregulation of the necroptosis marker receptor-interacting serine/threonine-protein kinase 3 (RIPK3) in renal cortical sections. Western blot analysis further demonstrated enhanced levels of phosphorylated RIPK3 in the kidneys of caspase-3-/- mice. In contrast, caspase-3-/- mice had less microvascular congestion and activation in the early and extension phases of AKI. In the long term (3 weeks after IRI), caspase-3-/- mice had reduced microvascular rarefaction and renal fibrosis, as well as decreased expression of α-smooth muscle actin and reduced collagen deposition within peritubular capillaries. Moreover, caspase-3-/- mice exhibited signs of reduced tubular ischemia, including lower tubular expression of hypoxia-inducible factor-1α and improved tubular injury scores.Conclusions These results establish the pivotal importance of caspase-3 in regulating microvascular endothelial cell apoptosis and renal fibrosis after IRI. These findings also demonstrate the predominant role of microvascular over tubular injury as a driver of progressive renal damage and fibrosis after IRI.
Keywords: acute kidney injury; apoptosis; caspase-3; ischemia-reperfusion.
Copyright © 2018 by the American Society of Nephrology.
Figures



Similar articles
-
Caspase-3-dependent peritubular capillary dysfunction is pivotal for the transition from acute to chronic kidney disease after acute ischemia-reperfusion injury.Am J Physiol Renal Physiol. 2021 Sep 1;321(3):F335-F351. doi: 10.1152/ajprenal.00690.2020. Epub 2021 Aug 2. Am J Physiol Renal Physiol. 2021. PMID: 34338031
-
Loss of receptor interacting protein kinases 3 and caspase-8 augments intrinsic apoptosis in tubular epithelial cell and promote kidney ischaemia-reperfusion injury.Nephrology (Carlton). 2019 Jun;24(6):661-669. doi: 10.1111/nep.13487. Epub 2019 Apr 29. Nephrology (Carlton). 2019. PMID: 30175514 Free PMC article.
-
Akt1 is involved in tubular apoptosis and inflammatory response during renal ischemia-reperfusion injury.Mol Biol Rep. 2020 Dec;47(12):9511-9520. doi: 10.1007/s11033-020-06021-1. Epub 2020 Nov 27. Mol Biol Rep. 2020. PMID: 33247386
-
Regulated necrosis in kidney ischemia-reperfusion injury.Kidney Int. 2019 Aug;96(2):291-301. doi: 10.1016/j.kint.2019.02.009. Epub 2019 Mar 7. Kidney Int. 2019. PMID: 31005270 Review.
-
Metabolic Flexibility and Innate Immunity in Renal Ischemia Reperfusion Injury: The Fine Balance Between Adaptive Repair and Tissue Degeneration.Front Immunol. 2020 Jul 7;11:1346. doi: 10.3389/fimmu.2020.01346. eCollection 2020. Front Immunol. 2020. PMID: 32733450 Free PMC article. Review.
Cited by
-
Microvascular disease in chronic kidney disease: the base of the iceberg in cardiovascular comorbidity.Clin Sci (Lond). 2020 Jun 26;134(12):1333-1356. doi: 10.1042/CS20200279. Clin Sci (Lond). 2020. PMID: 32542397 Free PMC article. Review.
-
Ganoderic Acids Prevent Renal Ischemia Reperfusion Injury by Inhibiting Inflammation and Apoptosis.Int J Mol Sci. 2021 Sep 23;22(19):10229. doi: 10.3390/ijms221910229. Int J Mol Sci. 2021. PMID: 34638569 Free PMC article.
-
Dynamic cellular changes in acute kidney injury caused by different ischemia time.iScience. 2023 Apr 11;26(5):106646. doi: 10.1016/j.isci.2023.106646. eCollection 2023 May 19. iScience. 2023. PMID: 37168554 Free PMC article.
-
Effect of fetuin-A on adenine-induced chronic kidney disease model in male rats.Iran J Basic Med Sci. 2023;26(5):511-516. doi: 10.22038/IJBMS.2023.66346.14584. Iran J Basic Med Sci. 2023. PMID: 37051102 Free PMC article.
-
Apoptotic cell death in disease-Current understanding of the NCCD 2023.Cell Death Differ. 2023 May;30(5):1097-1154. doi: 10.1038/s41418-023-01153-w. Epub 2023 Apr 26. Cell Death Differ. 2023. PMID: 37100955 Free PMC article. Review.
References
-
- Bedford M, Farmer C, Levin A, Ali T, Stevens P: Acute kidney injury and CKD: Chicken or egg? Am J Kidney Dis 59: 485–491, 2012 - PubMed
-
- Amdur RL, Chawla LS, Amodeo S, Kimmel PL, Palant CE: Outcomes following diagnosis of acute renal failure in U.S. veterans: Focus on acute tubular necrosis. Kidney Int 76: 1089–1097, 2009 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

