Possible Molecular Targets of Novel Ruthenium Complexes in Antiplatelet Therapy
- PMID: 29925802
- PMCID: PMC6032250
- DOI: 10.3390/ijms19061818
Possible Molecular Targets of Novel Ruthenium Complexes in Antiplatelet Therapy
Abstract
In oncotherapy, ruthenium (Ru) complexes are reflected as potential alternatives for platinum compounds and have been proved as encouraging anticancer drugs with high efficacy and low side effects. Cardiovascular diseases (CVDs) are mutually considered as the number one killer globally, and thrombosis is liable for the majority of CVD-related deaths. Platelets, an anuclear and small circulating blood cell, play key roles in hemostasis by inhibiting unnecessary blood loss of vascular damage by making blood clot. Platelet activation also plays a role in cancer metastasis and progression. Nevertheless, abnormal activation of platelets results in thrombosis under pathological settings such as the rupture of atherosclerotic plaques. Thrombosis diminishes the blood supply to the heart and brain resulting in heart attacks and strokes, respectively. While currently used anti-platelet drugs such as aspirin and clopidogrel demonstrate efficacy in many patients, they exert undesirable side effects. Therefore, the development of effective therapeutic strategies for the prevention and treatment of thrombotic diseases is a demanding priority. Recently, precious metal drugs have conquered the subject of metal-based drugs, and several investigators have motivated their attention on the synthesis of various ruthenium (Ru) complexes due to their prospective therapeutic values. Similarly, our recent studies established that novel ruthenium-based compounds suppressed platelet aggregation via inhibiting several signaling cascades. Our study also described the structure antiplatelet-activity relationship (SAR) of three newly synthesized ruthenium-based compounds. This review summarizes the antiplatelet activity of newly synthesized ruthenium-based compounds with their potential molecular mechanisms.
Keywords: antiplatelet; antithrombosis; ruthenium complex; signaling cascades.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
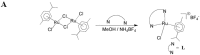
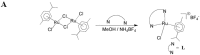

 inhibits;
inhibits;  blocks;
blocks;  decrease;
decrease;  increase.
increase.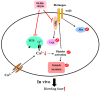
References
-
- Boucharaba A., Serre C.M., Gres S., Saulnier-Blache J.S., Bordet J.C., Guglielmi J., Clezardin P., Peyruchaud O. Platelet-derived lysophosphatidic acid supports the progression of osteolytic bone metastases in breast cancer. J. Clin. Investig. 2004;114:1714–1725. doi: 10.1172/JCI200422123. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

