α2- and β2-Adrenoreceptor-Mediated Efficacy of the Atypical Antidepressant Agomelatine Combined With Gabapentin to Suppress Allodynia in Neuropathic Rats With Ligated Infraorbital or Sciatic Nerve
- PMID: 29930510
- PMCID: PMC5999781
- DOI: 10.3389/fphar.2018.00587
α2- and β2-Adrenoreceptor-Mediated Efficacy of the Atypical Antidepressant Agomelatine Combined With Gabapentin to Suppress Allodynia in Neuropathic Rats With Ligated Infraorbital or Sciatic Nerve
Abstract
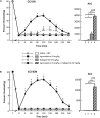
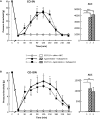
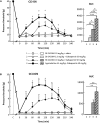
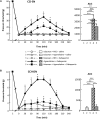
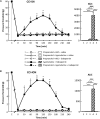
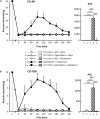
Previous data showed that neuropathic pain induced by mechanical lesion of peripheral nerves has specific characteristics and responds differently to alleviating drugs at cephalic versus extracephalic level. This is especially true for tricyclic antidepressants currently used for alleviating neuropathic pain in humans which are less effective against cephalic neuropathic pain. Whether this also applies to the antidepressant agomelatine, with its unique pharmacological properties as MT1/MT2 melatonin receptor agonist and 5-HT2B/5-HT2C serotonin receptor antagonist, has been investigated in two rat models of neuropathic pain. Acute treatments were performed 2 weeks after unilateral chronic constriction (ligation) injury to the sciatic nerve (CCI-SN) or the infraorbital nerve (CCI-ION), when maximal mechanical allodynia had developed in ipsilateral hindpaw or vibrissal pad, respectively, in Sprague-Dawley male rats. Although agomelatine (45 mg/kg i.p.) alone was inactive, co-treatment with gabapentin, at an essentially ineffective dose (50 mg/kg i.p.) on its own, produced marked anti-allodynic effects, especially in CCI-ION rats. In both CCI-SN and CCI-ION models, suppression of mechanical allodynia by 'agomelatine + gabapentin' could be partially mimicked by the combination of 5-HT2C antagonist (SB 242084) + gabapentin, but not by melatonin or 5-HT2B antagonist (RS 127445, LY 266097), alone or combined with gabapentin. In contrast, pretreatment by idazoxan, propranolol or the β2 antagonist ICI 118551 markedly inhibited the anti-allodynic effect of 'agomelatine + gabapentin' in both CCI-SN and CCI-ION rats, whereas pretreatment by the MT1/MT2 receptor antagonist S22153 was inactive. Altogether these data indicate that 'agomelatine + gabapentin' is a potent anti-allodynic combination at both cephalic and extra-cephalic levels, whose action implicates α2- and β2-adrenoreceptor-mediated noradrenergic neurotransmission.
Keywords: agomelatine; chronic constriction injury; gabapentin; infraorbital nerve; mechanical allodynia; neuropathic rats; sciatic nerve; α2- and β2-adrenoreceptors.
Figures


Similar articles
-
Agomelatine: a new opportunity to reduce neuropathic pain-preclinical evidence.Pain. 2017 Jan;158(1):149-160. doi: 10.1097/j.pain.0000000000000738. Pain. 2017. PMID: 27984527
-
Differential effects of calcitonin gene-related peptide receptor blockade by olcegepant on mechanical allodynia induced by ligation of the infraorbital nerve vs the sciatic nerve in the rat.Pain. 2012 Sep;153(9):1939-1948. doi: 10.1016/j.pain.2012.06.009. Epub 2012 Jul 15. Pain. 2012. PMID: 22795918
-
Effects of tapentadol on mechanical hypersensitivity in rats with ligatures of the infraorbital nerve versus the sciatic nerve.Eur J Pain. 2013 Jul;17(6):867-80. doi: 10.1002/j.1532-2149.2012.00259.x. Epub 2012 Dec 11. Eur J Pain. 2013. PMID: 23229959
-
Agomelatine: A Potential Multitarget Compound for Neurodevelopmental Disorders.Brain Sci. 2023 Apr 27;13(5):734. doi: 10.3390/brainsci13050734. Brain Sci. 2023. PMID: 37239206 Free PMC article. Review.
-
Profile of agomelatine and its potential in the treatment of generalized anxiety disorder.Neuropsychiatr Dis Treat. 2015 May 5;11:1149-55. doi: 10.2147/NDT.S67470. eCollection 2015. Neuropsychiatr Dis Treat. 2015. PMID: 25999720 Free PMC article. Review.
Cited by
-
PRECISE trial (Pain RElief Combination Intervention StratEgies): protocol for the clinical trial of a pregabalin-melatonin combination for fibromyalgia.BMJ Open. 2024 Jun 23;14(6):e087180. doi: 10.1136/bmjopen-2024-087180. BMJ Open. 2024. PMID: 38910006 Free PMC article.
-
The effectiveness of pregabalin with or without agomelatine in the treatment of chronic low back pain: a double-blind, placebo-controlled, randomized clinical trial.BMC Pharmacol Toxicol. 2022 Sep 14;23(1):70. doi: 10.1186/s40360-022-00612-3. BMC Pharmacol Toxicol. 2022. PMID: 36104745 Free PMC article. Clinical Trial.
-
Role of Melatonin in the Regulation of Pain.J Pain Res. 2020 Feb 7;13:331-343. doi: 10.2147/JPR.S228577. eCollection 2020. J Pain Res. 2020. PMID: 32104055 Free PMC article. Review.
References
-
- Auclair A. L., Cathala A., Sarrazin F., Depoortère R., Piazza P. V., Newman-Tancredi A., et al. (2010). The central serotonin2B receptor: a new pharmacological target to modulate the mesoaccumbens dopaminergic pathway activity. J. Neurochem. 114 1323–1332. 10.1111/j.1471-4159.2010.06848.x - DOI - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources

