Regulation of Gastric Lgr5+ve Cell Homeostasis by Bone Morphogenetic Protein (BMP) Signaling and Inflammatory Stimuli
- PMID: 29930977
- PMCID: PMC6009760
- DOI: 10.1016/j.jcmgh.2018.01.007
Regulation of Gastric Lgr5+ve Cell Homeostasis by Bone Morphogenetic Protein (BMP) Signaling and Inflammatory Stimuli
Abstract
Background & aims: Gastric Leucine-rich repeat-containing G-protein-coupled receptor 5 (Lgr5) cells exert important functions during injury and homeostasis. Bone morphogenetic protein (BMP) signaling regulates gastric inflammation and epithelial homeostasis. We investigated if BMP signaling controls the fate of Lgr5+ve cells during inflammation.
Methods: The H+/K+-adenosine triphosphatase β-subunit promoter was used to express the BMP inhibitor noggin (Nog) in the stomach (H+/K+-Nog mice). Inhibition of BMP signaling in Lgr5 cells was achieved by crossing Lgr5-EGFP-ires-CreERT2 (Lgr5-Cre) mice to mice with floxed alleles of BMP receptor 1A (Lgr5-Cre;Bmpr1aflox/flox mice). Lgr5/GFP+ve cells were isolated using flow cytometry. Lineage tracing studies were conducted by crossing Lgr5-Cre mice to mice that express Nog and tdTomato (Lgr5-Cre;H+/K+-Nog;Rosa26-tdTom). Infection with Helicobacter felis was used to induce inflammation. Morphology of the mucosa was analyzed by H&E staining. Distribution of H+/K+-adenosine triphosphatase-, IF-, Ki67-, CD44-, CD44v9-, and bromodeoxyuridine-positive cells was analyzed by immunostaining. Expression of neck and pit cell mucins was determined by staining with the lectins Griffonia (Bandeiraea) simplicifolia lectin II and Ulex europaeus agglutinin 1, respectively. Id1, Bmpr1a, Lgr5, c-Myc, and Cd44 messenger RNAs were measured by quantitative reverse-transcription polymerase chain reaction.
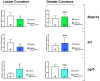
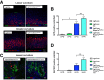
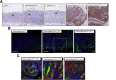
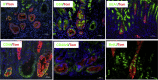
Results: Lgr5-Cre;Bmpr1aflox/flox mice showed diminished expression of Bmpr1a in Lgr5/GFP+ve cells. Infection of Lgr5-Cre;Bmpr1aflox/flox mice with H felis led to enhanced inflammation, increased cell proliferation, parietal cell loss, and to the development of metaplasia and dysplasia. Infected Lgr5-Cre;H+/K+-Nog;Rosa26-tdTom mice, but not control mice, showed the presence of tomato+ve glands lining the lesser curvature that stained positively with Griffonia (Bandeiraea) simplicifolia lectin II and Ulex europaeus agglutinin 1, and with anti-IF, -CD44, -CD44v9, and -bromodeoxyuridine antibodies.
Conclusions: Inflammation and inhibition of BMP signaling activate Lgr5+ve cells, which give rise to metaplastic, dysplastic, proliferating lineages that express markers of mucus neck and zymogenic cell differentiation.
Keywords: ATPase, adenosine triphosphatase; BMP, bone morphogenetic protein; BrdU, bromodeoxyuridine; Chief Cells; Differentiation; Dysplasia; EGFP, enhanced green fluorescent protein; ERK, extracellular signal–regulated kinase; GFP, green fluorescent protein; GSII, Griffonia (Bandeiraea) simplicifolia lectin II; H/K-nog, H/K-noggin; HBSS, Hank's balanced salt solution; IF, intrinsic factor; Metaplasia; QRT-PCR, quantitative reverse-transcription polymerase chain reaction; SPEM, spasmolytic polypeptide expressing metaplasia; TFF2, Trefoil factor 2; mRNA, messenger RNA.
Figures







References
-
- Peek R.M., Jr. IV. Helicobacter pylori strain-specific activation of signal transduction cascades related to gastric inflammation. Am J Physiol Gastrointest Liver Physiol. 2001;280:G525–G530. - PubMed
-
- Nomura S., Baxter T., Yamaguchi H., Leys C., Vartapetian A.B., Fox J.G., Lee J.R., Wang T.C., Goldenring J.R. Spasmolytic polypeptide expressing metaplasia to preneoplasia in H. felis-infected mice. Gastroenterology. 2004;127:582–594. - PubMed
-
- Zavros Y., Eaton K.A., Kang W., Rathinavelu S., Katukuri V., Kao J.Y., Samuelson L.C., Merchant J.L. Chronic gastritis in the hypochlorhydric gastrin-deficient mouse progresses to adenocarcinoma. Oncogene. 2005;24:2354–2366. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

