Proteasome inhibitors: structure and function
- PMID: 29935898
- PMCID: PMC6020165
- DOI: 10.1053/j.seminoncol.2018.01.004
Proteasome inhibitors: structure and function
Abstract
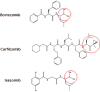
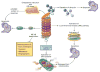
Since 2003, the US Food and Drug Administration approval of bortezomib, a proteasome inhibitor, has changed the management of hematologic malignancies and dramatically improved outcomes for patients with multiple myeloma and mantle cell lymphoma. Since that time, two additional proteasome inhibitors (carfilzomib and ixazomib) have been approved, with other agents and combinations currently under investigation. Proteasomes degrade ubiquitinated proteins or substrates through the ubiquitin-proteasome pathway, a pathway that is utilized in multiple myeloma because of the high protein turnover with immunoglobulin production. Proteasome inhibitors exploit dependence on this pathway, halting protein degradation that ultimately results in apoptosis and cell death. Here we will discuss the structure of the proteasome and the mechanisms of action for proteasome inhibitors to further understand their role in hematologic malignancies.
Keywords: bortezomib; multiple myeloma; proteasome inhibitors; ubiquitin-proteasome pathway.
Published by Elsevier Inc.
Figures
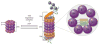
References
-
- King RW, Deshaies RJ, Peters J-M, Kirschner MW. How Proteolysis Drives the Cell Cycle. Science. 1996;274(5293):1652–9. - PubMed
-
- Adams J. The proteasome: structure, function, and role in the cell. Cancer Treatment Reviews. 2003;29(Supplement 1):3–9. - PubMed
-
- Palombella VJ, Rando OJ, Goldberg AL, Maniatis T. The ubiquitin-proteasome pathway is required for processing the NF-kappa B1 precursor protein and the activation of NF-kappa B. Cell. 1994;78(5):773–85. - PubMed
-
- Hideshima T, Chauhan D, Richardson P, Mitsiades C, Mitsiades N, Hayashi T, et al. NF-κB as a Therapeutic Target in Multiple Myeloma. Journal of Biological Chemistry. 2002;277(19):16639–47. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

