Evaluation of optical imaging agents in a fluorescence-guided surgical model of head and neck cancer
- PMID: 29937175
- PMCID: PMC6072276
- DOI: 10.1016/j.suronc.2018.04.004
Evaluation of optical imaging agents in a fluorescence-guided surgical model of head and neck cancer
Abstract
Background: Tumor proliferation often occurs from pathologic receptor upregulation. These receptors provide unique targets for near-infrared (NIR) probes that have fluorescence-guided surgery (FGS) applications. We demonstrate the use of three smart-targeted probes in a model of head and neck squamous cell carcinoma.
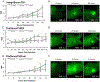
Methods: A dose escalation study was performed using IntegriSense750, ProSense750EX, and ProSense750FAST in mice (n = 5) bearing luciferase-positive SCC-1 flank xenograft tumors. Whole body fluorescence imaging was performed serially after intravenous injection using commercially available open-field (LUNA, Novadaq, Canada) and closed-field NIR systems (Pearl, LI-COR, Lincoln, NE). An ex vivo, whole-body biodistribution was conducted. Lastly, FGS was performed with IntegriSense750 to demonstrate orthotopic and metastatic disease localization.
Results: Disease fluorescence delineation was assessed by tumor-to-background fluorescence ratios (TBR). Peak TBR values were 3.3 for 1 nmol ProSense750EX, 5.5 for 6 nmol ProSense750FAST, and 10.8 for 4 nmol IntegriSense750 at 5.5, 3, and 4 d post administration, respectively. Agent utility is unique: ProSense750FAST provides sufficient contrast quickly (TBR: 1.5, 3 h) while IntegriSense750 produces strong (TBR: 10.8) contrast with extended administration-to-resection time (96 h). IntegriSense750 correctly identified all diseased nodes in situ during exploratory surgeries. Ex vivo, whole-body biodistribution was assessed by tumor-to-tissue fluorescence ratios (TTR). Agents provided sufficient fluorescence contrast to discriminate disease from background, TTR>1. IntegriSense750 was most robust in neural tissue (TTR: 64) while ProSense750EX was superior localizing disease against lung tissue (TBR: 13).
Conclusion: All three agents appear effective for FGS.
Keywords: Fluorescence imaging; Head and neck cancer; Optical guided surgery; Surgical oncology.
Copyright © 2018 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Figures





Similar articles
-
Evaluation of fluorescence-guided surgery agents in a murine model of soft tissue fibrosarcoma.J Surg Oncol. 2018 May;117(6):1179-1187. doi: 10.1002/jso.24950. Epub 2017 Dec 28. J Surg Oncol. 2018. PMID: 29284070
-
uPAR-targeted optical near-infrared (NIR) fluorescence imaging and PET for image-guided surgery in head and neck cancer: proof-of-concept in orthotopic xenograft model.Oncotarget. 2017 Feb 28;8(9):15407-15419. doi: 10.18632/oncotarget.14282. Oncotarget. 2017. PMID: 28039488 Free PMC article.
-
Optimal Dosing Strategy for Fluorescence-Guided Surgery with Panitumumab-IRDye800CW in Head and Neck Cancer.Mol Imaging Biol. 2020 Feb;22(1):156-164. doi: 10.1007/s11307-019-01358-x. Mol Imaging Biol. 2020. PMID: 31054001 Free PMC article.
-
Intraoperative Fluorescence-Guided Surgery in Head and Neck Squamous Cell Carcinoma.Laryngoscope. 2021 Mar;131(3):529-534. doi: 10.1002/lary.28822. Epub 2020 Jul 3. Laryngoscope. 2021. PMID: 33593036 Review.
-
Infrared Fluorescence-guided Surgery for Tumor and Metastatic Lymph Node Detection in Head and Neck Cancer.Radiol Imaging Cancer. 2024 Jul;6(4):e230178. doi: 10.1148/rycan.230178. Radiol Imaging Cancer. 2024. PMID: 38940689 Free PMC article. Review.
Cited by
-
Pressure-enhanced sensing of tissue oxygenation via endogenous porphyrin: Implications for dynamic visualization of cancer in surgery.Proc Natl Acad Sci U S A. 2024 Aug 20;121(34):e2405628121. doi: 10.1073/pnas.2405628121. Epub 2024 Aug 14. Proc Natl Acad Sci U S A. 2024. PMID: 39141355 Free PMC article.
-
Advances in Intraoperative Flow Cytometry.Int J Mol Sci. 2022 Nov 3;23(21):13430. doi: 10.3390/ijms232113430. Int J Mol Sci. 2022. PMID: 36362215 Free PMC article. Review.
-
In vivo visualization and molecular targeting of the cardiac conduction system.J Clin Invest. 2022 Oct 17;132(20):e156955. doi: 10.1172/JCI156955. J Clin Invest. 2022. PMID: 35951416 Free PMC article.
-
[18F]Fluciclovine-PET Guided Salvage Lymph Node Dissection Following Radical Prostatectomy.Urology. 2019 Oct;132:28-32. doi: 10.1016/j.urology.2019.05.022. Epub 2019 May 29. Urology. 2019. PMID: 31152764 Free PMC article. No abstract available.
References
-
- Twaalfhoven FC, Peters AA, Trimbos JB, Hermans J, Fleuren GJ. The accuracy of frozen section diagnosis of ovarian tumors. Gynecol Oncol. 1991;41(3):189–192. - PubMed
-
- Stummer W, Pichlmeier U, Meinel T, et al. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol. 2006;7(5):392–401. - PubMed
-
- Stummer W, Novotny A, Stepp H, Goetz C, Bise K, Reulen HJ. Fluorescence-guided resection of glioblastoma multiforme by using 5-aminolevulinic acid-induced porphyrins: a prospective study in 52 consecutive patients. J Neurosurg. 2000;93(6):1003–1013. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

