Monocyte Subsets Have Distinct Patterns of Tetraspanin Expression and Different Capacities to Form Multinucleate Giant Cells
- PMID: 29937768
- PMCID: PMC6002745
- DOI: 10.3389/fimmu.2018.01247
Monocyte Subsets Have Distinct Patterns of Tetraspanin Expression and Different Capacities to Form Multinucleate Giant Cells
Abstract
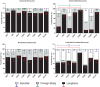
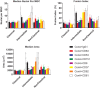
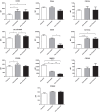
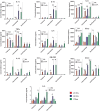
Monocytes are able to undergo homotypic fusion to produce different types of multinucleated giant cells, such as Langhans giant cells in response to M. tuberculosis infection or foreign body giant cells in response to implanted biomaterials. Monocyte fusion is highly coordinated and complex, with various soluble, intracellular, and cell-surface components mediating different stages of the process. Tetraspanins, such as CD9, CD63, and CD81, are known to be involved in cell:cell fusion and have been suggested to play a role in regulating homotypic monocyte fusion. However, peripheral human monocytes are not homogenous: they exist as a heterogeneous population consisting of three subsets, classical (CD14++CD16-), intermediate (CD14++CD16+), and non-classical (CD14+CD16+), at steady state. During infection with mycobacteria, the circulating populations of intermediate and non-classical monocytes increase, suggesting they may play a role in the disease outcome. Human monocytes were separated into subsets and then induced to fuse using concanavalin A. The intermediate monocytes were able to fuse faster and form significantly larger giant cells than the other subsets. When antibodies targeting tetraspanins were added, the intermediate monocytes responded to anti-CD63 by forming smaller giant cells, suggesting an involvement of tetraspanins in fusion for at least this subset. However, the expression of fusion-associated tetraspanins on monocyte subsets did not correlate with the extent of fusion or with the inhibition by tetraspanin antibody. We also identified a CD9High and a CD9Low monocyte population within the classical subset. The CD9High classical monocytes expressed higher levels of tetraspanin CD151 compared to CD9Low classical monocytes but the CD9High classical subset did not exhibit greater potential to fuse and the role of these cells in immunity remains unknown. With the exception of dendrocyte-expressed seven transmembrane protein, which was expressed at higher levels on the intermediate monocyte subset, the expression of fusion-related proteins between the subsets did not clearly correlate with their ability to fuse. We also did not observe any clear correlation between giant cell formation and the expression of pro-inflammatory or fusogenic cytokines. Although tetraspanin expression appears to be important for the fusion of intermediate monocytes, the control of multinucleate giant cell formation remains obscure.
Keywords: cd9; fusion; monocyte; monocyte subsets; tetraspanin.
Figures


Similar articles
-
Characterization of tetraspanins CD9, CD53, CD63, and CD81 in monocytes and macrophages in HIV-1 infection.J Leukoc Biol. 2013 Jun;93(6):913-20. doi: 10.1189/jlb.0812391. Epub 2013 Apr 9. J Leukoc Biol. 2013. PMID: 23570947
-
Distinct roles for tetraspanins CD9, CD63 and CD81 in the formation of multinucleated giant cells.Immunology. 2009 Jun;127(2):237-48. doi: 10.1111/j.1365-2567.2008.02945.x. Immunology. 2009. PMID: 19489128 Free PMC article.
-
A role for tetraspanin proteins in regulating fusion induced by Burkholderia thailandensis.Med Microbiol Immunol. 2020 Aug;209(4):473-487. doi: 10.1007/s00430-020-00670-6. Epub 2020 Apr 6. Med Microbiol Immunol. 2020. PMID: 32253503 Free PMC article.
-
Monocyte Subsets: Phenotypes and Function in Tuberculosis Infection.Front Immunol. 2018 Jul 30;9:1726. doi: 10.3389/fimmu.2018.01726. eCollection 2018. Front Immunol. 2018. PMID: 30105020 Free PMC article. Review.
-
Pattern of human monocyte subpopulations in health and disease.Scand J Immunol. 2020 Jul;92(1):e12883. doi: 10.1111/sji.12883. Epub 2020 May 5. Scand J Immunol. 2020. PMID: 32243617 Review.
Cited by
-
Tetraspanin CD37 regulates platelet hyperreactivity and thrombosis.Cardiovasc Res. 2025 Jun 12;121(6):943-956. doi: 10.1093/cvr/cvaf051. Cardiovasc Res. 2025. PMID: 40126944
-
Effects of Mycobacterium vaccae vaccine in a mouse model of tuberculosis: protective action and differentially expressed genes.Mil Med Res. 2020 Jun 3;7(1):25. doi: 10.1186/s40779-020-00258-4. Mil Med Res. 2020. PMID: 32493477 Free PMC article.
-
The Implant-Induced Foreign Body Response Is Limited by CD13-Dependent Regulation of Ubiquitination of Fusogenic Proteins.J Immunol. 2024 Feb 15;212(4):663-676. doi: 10.4049/jimmunol.2300688. J Immunol. 2024. PMID: 38149920 Free PMC article.
-
Learning from Monocyte-Macrophage Fusion and Multinucleation: Potential Therapeutic Targets for Osteoporosis and Rheumatoid Arthritis.Int J Mol Sci. 2020 Aug 20;21(17):6001. doi: 10.3390/ijms21176001. Int J Mol Sci. 2020. PMID: 32825443 Free PMC article. Review.
-
Methamphetamine induces transcriptional changes in cultured HIV-infected mature monocytes that may contribute to HIV neuropathogenesis.Front Immunol. 2022 Aug 18;13:952183. doi: 10.3389/fimmu.2022.952183. eCollection 2022. Front Immunol. 2022. PMID: 36059515 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

