Estimation of fitness from energetics and life-history data: An example using mussels
- PMID: 29938052
- PMCID: PMC6010765
- DOI: 10.1002/ece3.4004
Estimation of fitness from energetics and life-history data: An example using mussels
Abstract
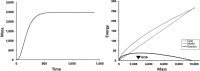
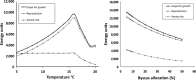
Changing environments have the potential to alter the fitness of organisms through effects on components of fitness such as energy acquisition, metabolic cost, growth rate, survivorship, and reproductive output. Organisms, on the other hand, can alter aspects of their physiology and life histories through phenotypic plasticity as well as through genetic change in populations (selection). Researchers examining the effects of environmental variables frequently concentrate on individual components of fitness, although methods exist to combine these into a population level estimate of average fitness, as the per capita rate of population growth for a set of identical individuals with a particular set of traits. Recent advances in energetic modeling have provided excellent data on energy intake and costs leading to growth, reproduction, and other life-history parameters; these in turn have consequences for survivorship at all life-history stages, and thus for fitness. Components of fitness alone (performance measures) are useful in determining organism response to changing conditions, but are often not good predictors of fitness; they can differ in both form and magnitude, as demonstrated in our model. Here, we combine an energetics model for growth and allocation with a matrix model that calculates population growth rate for a group of individuals with a particular set of traits. We use intertidal mussels as an example, because data exist for some of the important energetic and life-history parameters, and because there is a hypothesized energetic trade-off between byssus production (affecting survivorship), and energy used for growth and reproduction. The model shows exactly how strong this trade-off is in terms of overall fitness, and it illustrates conditions where fitness components are good predictors of actual fitness, and cases where they are not. In addition, the model is used to examine the effects of environmental change on this trade-off and on both fitness and on individual fitness components.
Keywords: climate change; energetics; fitness; intertidal; invertebrate; life‐history; mussels.
Figures



References
-
- Angilletta, M. J. , & Dunham, A. E. (2003). The temperature‐size rule in ectotherms: Simple evolutionary explanations may not be general. The American Naturalist, 162, 332–342. https://doi.org/10.1086/377187 - DOI - PubMed
-
- Angilletta, M. J. , Steury, T. D. , & Sears, M. W. (2004). Temperature, growth rate, and body size in ectotherms: Fitting pieces of a life‐history puzzle. Integrative and Comparative Biology, 44, 498–509. https://doi.org/10.1093/icb/44.6.498 - DOI - PubMed
-
- Arnold, S. J. , & Wade, M. J. (1984a). On the measurement of natural and sexual selection: Theory. Evolution, 38, 709–719. https://doi.org/10.1111/j.1558-5646.1984.tb00344.x - DOI - PubMed
-
- Arnold, S. J. , & Wade, M. J. (1984b). On the measurement of natural and sexual selection: Applications. Evolution, 38, 720–734. https://doi.org/10.1111/j.1558-5646.1984.tb00345.x - DOI - PubMed
-
- Babarro, J. M. F. , & Carrington, E. (2013). Attachment strength of the mussel Mytilus galloprovincialis: Effect of habitat and body size. Journal of Experimental Marine Biology and Ecology, 443, 188–196. https://doi.org/10.1016/j.jembe.2013.02.035 - DOI
LinkOut - more resources
Full Text Sources
Other Literature Sources

