Late administration of high-frequency electrical stimulation increases nerve regeneration without aggravating neuropathic pain in a nerve crush injury
- PMID: 29940857
- PMCID: PMC6020201
- DOI: 10.1186/s12868-018-0437-9
Late administration of high-frequency electrical stimulation increases nerve regeneration without aggravating neuropathic pain in a nerve crush injury
Abstract
Background: High-frequency transcutaneous neuromuscular electrical nerve stimulation (TENS) is currently used for the administration of electrical current in denervated muscle to alleviate muscle atrophy and enhance motor function; however, the time window (i.e. either immediate or delayed) for achieving benefit is still undetermined. In this study, we conducted an intervention of sciatic nerve crush injury using high-frequency TENS at different time points to assess the effect of motor and sensory functional recovery.
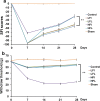
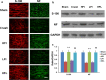
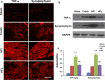
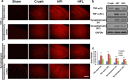
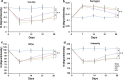
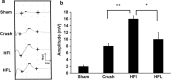
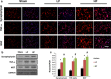
Results: Animals with left sciatic nerve crush injury received TENS treatment starting immediately after injury or 1 week later at a high frequency(100 Hz) or at a low frequency (2 Hz) as a control. In SFI gait analysis, either immediate or late admission of high-frequency electrical stimulation exerted significant improvement compared to either immediate or late administration of low-frequency electrical stimulation. In an assessment of allodynia, immediate high frequency electrical stimulation caused a significantly decreased pain threshold compared to late high-frequency or low-frequency stimulation at immediate or late time points. Immunohistochemistry staining and western blot analysis of S-100 and NF-200 demonstrated that both immediate and late high frequency electrical stimulation showed a similar effect; however the effect was superior to that achieved with low frequency stimulation. Immediate high frequency electrical stimulation resulted in significant expression of TNF-α and synaptophysin in the dorsal root ganglion, somatosensory cortex, and hippocampus compared to late electrical stimulation, and this trend paralleled the observed effect on somatosensory evoked potential. The CatWalk gait analysis also showed that immediate electrical stimulation led to a significantly high regularity index. In primary dorsal root ganglion cells culture, high-frequency electrical stimulation also exerted a significant increase in expression of TNF-α, synaptophysin, and NGF in accordance with the in vivo results.
Conclusion: Immediate or late transcutaneous high-frequency electrical stimulation exhibited the potential to stimulate the motor nerve regeneration. However, immediate electrical stimulation had a predilection to develop neuropathic pain. A delay in TENS initiation appears to be a reasonable approach for nerve repair and provides the appropriate time profile for its clinical application.
Keywords: Dorsal root ganglion cell; Nerve regeneration; Neuropathic pain; Transcutaneous electrical stimulation.
Figures
Similar articles
-
Effectiveness of electrical stimulation on nerve regeneration after crush injury: Comparison between invasive and non-invasive stimulation.PLoS One. 2020 May 26;15(5):e0233531. doi: 10.1371/journal.pone.0233531. eCollection 2020. PLoS One. 2020. PMID: 32453807 Free PMC article.
-
High- and low-frequency transcutaneous electrical nerve stimulation delay sciatic nerve regeneration after crush lesion in the mouse.J Peripher Nerv Syst. 2008 Mar;13(1):71-80. doi: 10.1111/j.1529-8027.2008.00160.x. J Peripher Nerv Syst. 2008. PMID: 18346233
-
Feasibility of Human Amniotic Fluid Derived Stem Cells in Alleviation of Neuropathic Pain in Chronic Constrictive Injury Nerve Model.PLoS One. 2016 Jul 21;11(7):e0159482. doi: 10.1371/journal.pone.0159482. eCollection 2016. PLoS One. 2016. PMID: 27441756 Free PMC article.
-
Nerve stimulation and neuromodulation for painful nerves: a narrative review.Int Orthop. 2025 May;49(5):989-996. doi: 10.1007/s00264-025-06498-0. Epub 2025 Mar 20. Int Orthop. 2025. PMID: 40111452 Review.
-
Transcutaneous Electrical Nerve Stimulation in Relieving Neuropathic Pain: Basic Mechanisms and Clinical Applications.Curr Pain Headache Rep. 2020 Feb 18;24(4):14. doi: 10.1007/s11916-020-0846-1. Curr Pain Headache Rep. 2020. PMID: 32072323 Review.
Cited by
-
Transcutaneous Electrical Nerve Stimulation in Rodent Models of Neuropathic Pain: A Meta-Analysis.Front Neurosci. 2022 Jan 31;16:831413. doi: 10.3389/fnins.2022.831413. eCollection 2022. Front Neurosci. 2022. PMID: 35173577 Free PMC article.
-
[Progress of electrical stimulation to promote peripheral nerve regeneration].Lin Chuang Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2024 May;38(5):411-415;420. doi: 10.13201/j.issn.2096-7993.2024.05.012. Lin Chuang Er Bi Yan Hou Tou Jing Wai Ke Za Zhi. 2024. PMID: 38686479 Free PMC article. Review. Chinese.
-
CatWalk XT gait parameters: a review of reported parameters in pre-clinical studies of multiple central nervous system and peripheral nervous system disease models.Front Behav Neurosci. 2023 Jun 7;17:1147784. doi: 10.3389/fnbeh.2023.1147784. eCollection 2023. Front Behav Neurosci. 2023. PMID: 37351154 Free PMC article. Review.
-
Efficiency of Direct Transcutaneous Electroneurostimulation of the Median Nerve in the Regression of Residual Neurological Symptoms after Carpal Tunnel Decompression Surgery.Biomedicines. 2023 Aug 27;11(9):2396. doi: 10.3390/biomedicines11092396. Biomedicines. 2023. PMID: 37760837 Free PMC article.
-
Modulation of Aryl Hydrocarbon Receptor Expression Alleviated Neuropathic Pain in a Chronic Constriction Nerve Injury Animal Model.Int J Mol Sci. 2022 Sep 24;23(19):11255. doi: 10.3390/ijms231911255. Int J Mol Sci. 2022. PMID: 36232555 Free PMC article.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

