Efficacy and safety of sofosbuvir-based antiviral therapy to treat hepatitis C virus infection after kidney transplantation
- PMID: 29942507
- PMCID: PMC6007709
- DOI: 10.1093/ckj/sfx112
Efficacy and safety of sofosbuvir-based antiviral therapy to treat hepatitis C virus infection after kidney transplantation
Abstract
Background: The objectives of this pilot study were to assess the efficacy and safety of an interferon-free sofosbuvir and ribavirin combination regimen to treat chronic hepatitis C virus (HCV) infection in kidney transplant recipients and to study the impact of sofosbuvir on calcineurin inhibitor (CNI) drug levels.
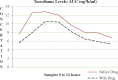
Methods: A total of 10 kidney transplant recipients with chronic HCV infection were included in the study. All received sofosbuvir and ribavirin combination therapy. The virological response to therapy and the adverse effects of the drugs were studied. The area under the curve (AUC) and pharmacokinetic data of levels of CNI were compared while the patients were receiving sofosbuvir and ribavirin drugs and when they were no longer on these drugs.
Results: In all, 9 of 10 patients (90%) achieved rapid virological response (RVR) with undetectable HCV RNA at 4 weeks and the remaining patient achieved undetectable HCV RNA at 8 weeks. A sustained virological response was seen at 3, 6 and 12 months and was maintained in all 10 patients (100%). The important aspect of the study is the effect of treatment with the sofosbuvir-ribavirin combination regimen on the CNI AUC levels, which resulted in a reduction in the CNI AUC. While used as part of triple-drug immunosuppression, no change in the dose of CNI (tacrolimus and cyclosporine) was required based on measurement of C0 levels.
Conclusions: The sofosbuvir and ribavirin combination therapy is effective and safe to treat HCV infection in the post-renal transplant setting. There is a need for close CNI level monitoring while these patients are on sofosbuvir therapy. With therapy and viral clearance, there could be reduction in CNI levels due to increased clearance of CNI drugs, which is shown by the AUC measurements. This could be important for patients at high risk for rejection.
Keywords: CNI drugs; hepatitis C virus; kidney transplant; ribavirin; sofosbuvir.
Figures
Similar articles
-
Efficacy and Safety of Sofosbuvir-Based Antiviral Therapy to Treat Hepatitis C Virus Infection After Kidney Transplantation.Am J Transplant. 2016 May;16(5):1474-9. doi: 10.1111/ajt.13518. Epub 2015 Nov 20. Am J Transplant. 2016. PMID: 26587971
-
Sofosbuvir (Sovaldi°). Active against hepatitis C virus, but evaluation is incomplete.Prescrire Int. 2015 Jan;24(156):5-10. Prescrire Int. 2015. PMID: 25734194
-
Safety and efficacy of protease inhibitors to treat hepatitis C after liver transplantation: a multicenter experience.J Hepatol. 2014 Jan;60(1):78-86. doi: 10.1016/j.jhep.2013.08.018. Epub 2013 Aug 29. J Hepatol. 2014. PMID: 23994384
-
Treatment with sofosbuvir and ledipasvir without ribavirin for 12 weeks is highly effective for recurrent hepatitis C virus genotype 1b infection after living donor liver transplantation: a Japanese multicenter experience.J Gastroenterol. 2017 Aug;52(8):986-991. doi: 10.1007/s00535-017-1310-9. Epub 2017 Jan 30. J Gastroenterol. 2017. PMID: 28138756
-
Hepatitis C treatment from "response-guided" to "resource-guided" therapy in the transition era from interferon-containing to interferon-free regimens.J Gastroenterol Hepatol. 2017 Aug;32(8):1436-1442. doi: 10.1111/jgh.13747. J Gastroenterol Hepatol. 2017. PMID: 28124463 Review.
Cited by
-
Direct-acting Antivirals for the Treatment of Kidney Transplant Patients With Chronic Hepatitis C Virus Infection in Spain: A Long-term Prospective Observational Study.Transplant Direct. 2019 Nov 18;5(12):e510. doi: 10.1097/TXD.0000000000000954. eCollection 2019 Dec. Transplant Direct. 2019. PMID: 32095505 Free PMC article.
-
Coincidence of HCV and chronic kidney disease-a systematic review and meta-analysis.BMC Public Health. 2024 Oct 16;24(1):2842. doi: 10.1186/s12889-024-20331-0. BMC Public Health. 2024. PMID: 39415151 Free PMC article.
-
A Systematic Review of Direct-Acting Antivirals for Hepatitis C in Advanced CKD.Kidney Int Rep. 2022 Dec 1;8(2):240-253. doi: 10.1016/j.ekir.2022.11.008. eCollection 2023 Feb. Kidney Int Rep. 2022. PMID: 36815114 Free PMC article.
-
Opioid epidemic and liver disease.JHEP Rep. 2019 Jul 9;1(3):240-255. doi: 10.1016/j.jhepr.2019.06.006. eCollection 2019 Sep. JHEP Rep. 2019. PMID: 32039374 Free PMC article. Review.
-
A cohort study on the relationship between education level and high-risk population of stroke.Ibrain. 2021 Sep 28;7(3):181-191. doi: 10.1002/j.2769-2795.2021.tb00082.x. eCollection 2021 Sep. Ibrain. 2021. PMID: 37786801 Free PMC article.
References
-
- Kamar N, Ribes D, Izopet J. et al. Treatment of hepatitis C virus infection (HCV) after renal transplantation: implications for HCV-positive dialysis patients awaiting a kidney transplant. Transplantation 2006; 82: 853–856 - PubMed
-
- Morales JM, Campistol JM.. Transplantation in the patient with hepatitis C. J Am Soc Nephrol 2000; 11:1343–1353 - PubMed
-
- Fabrizi F, Martin P, Ponticelli C.. Hepatitis C virus infection and renal transplantation. Am J Kidney Dis 2001; 38:919–934 - PubMed
-
- Fabrizi F, Dixit V, Messa P. et al. Antiviral therapy (pegylated interferon and ribavirin) of hepatitis C in dialysis patients: metaanalysis of clinical studies. J Viral Hepat 2014; 21: 681–689 - PubMed
-
- Rostaing L, Izopet J, Baron E. et al. Treatment of chronic hepatitis C with recombinant interferon alpha in kidney transplant recipients. Transplantation 1995; 59:1426–1431 - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources