Real-Life Clinical Effectiveness of Razumab® (the World's First Biosimilar of Ranibizumab) in Retinal Vein Occlusion: A Subgroup Analysis of the Pooled Retrospective RE-ENACT Study
- PMID: 29945143
- PMCID: PMC6390449
- DOI: 10.1159/000488602
Real-Life Clinical Effectiveness of Razumab® (the World's First Biosimilar of Ranibizumab) in Retinal Vein Occlusion: A Subgroup Analysis of the Pooled Retrospective RE-ENACT Study
Abstract
Background: This subgroup analysis of the RE-ENACT study evaluates the effectiveness of Razumab® (the world's first biosimilar of ranibizumab by Intas Pharmaceuticals Ltd.) in Indian patients with retinal vein occlusion (RVO).
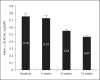
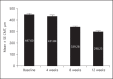
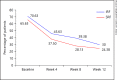
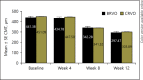
Methods: The data on patients with RVO who had received ≥3 injections of Razumab® between January and August 2016 were analyzed. Endpoints were: improvement in best corrected visual acuity (BCVA), and a decrease in central macular thickness (CMT), intraretinal fluid (IRF), and subretinal fluid (SRF) from baseline at weeks 4, 8, and 12.
Results: Of 160 patients, the majority (61.87%) were men. The mean (±SE) BCVA improved from baseline (0.76 ± 0.04) to week 4 (0.73 ± 0.03; p = 0.0656), which attained significance at week 8 (0.55 ± 0.02; p < 0.0001) and week 12 (0.47 ± 0.02; p < 0.0001). The mean (±SE) CMT significantly decreased from baseline (447.60 ± 10.91 μm) to week 4 (431.84 ± 10.92 μm; p = 0.0028), week 8 (339.28 ± 8.12 μm; p < 0.0001), and week 12 (298.23 ± 6.68 μm; p < 0.0001). The proportion of patients with IRF and SRF significantly (p < 0.0001) decreased from baseline to weeks 4, 8, and 12 (IRF: from 70.63 to 45.63, 39.38, and 30.00%, respectively; SRF: from 65.63 to 37.50, 28.13, and 24.38%, respectively). A subgroup analysis of branch RVO and central RVO showed similar results. No new safety concerns were observed.
Conclusion: Razumab® (biosimilar of ranibizumab) effectively improved visual acuity and disease outcomes in patients with RVO in a real-world setting with no new safety concerns.
Keywords: Biosimilar; Ranibizumab; Razumab®; Retinal vein occlusion.
The Author(s). Published by S. Karger AG, Basel.
Figures


References
-
- Mitchell P, Smith W, Chang A. Prevalence and associations of retinal vein occlusion in Australia. The Blue Mountains Eye Study. Arch Ophthalmol. 1996;114:1243–1247. - PubMed
-
- Kolar P. Definition and classification of retinal vein occlusion. Int J Ophthalm Res. 2016;2:124–129.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

