Growing cell walls show a gradient of elastic strain across their layers
- PMID: 29945239
- PMCID: PMC6093493
- DOI: 10.1093/jxb/ery237
Growing cell walls show a gradient of elastic strain across their layers
Erratum in
-
Erratum: Growing cell walls show a gradient of elastic strain across their layers.J Exp Bot. 2018 Sep 14;69(20):4987. doi: 10.1093/jxb/ery280. J Exp Bot. 2018. PMID: 30107529 Free PMC article. No abstract available.
Abstract
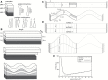
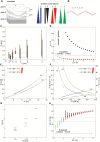
The relatively thick primary walls of epidermal and collenchyma cells often form waviness on the surface that faces the protoplast when they are released from the tensile in-plane stress that operates in situ. This waviness is a manifestation of buckling that results from the heterogeneity of the elastic strain across the wall. In this study, this heterogeneity was confirmed by the spontaneous bending of isolated wall fragments that were initially flat. We combined the empirical data on the formation of waviness in growing cell walls with computations of the buckled wall shapes. We chose cylindrical-shaped organs with a high degree of longitudinal tissue stress because in such organs the surface deformation that accompanies the removal of the stress is strongly anisotropic and leads to the formation of waviness in which wrinkles on the inner wall surface are always transverse to the organ axis. The computations showed that the strain heterogeneity results from individual or overlaid gradients of pre-stress and stiffness across the wall. The computed wall shapes depend on the assumed wall thickness and mechanical gradients. Thus, a quantitative analysis of the wall waviness that forms after stress removal can be used to assess the mechanical heterogeneity of the cell wall.
Figures
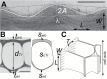
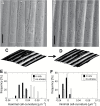
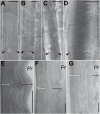
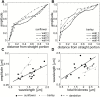

References
-
- Abasolo W, Eder M, Yamauchi K, et al. 2009. Pectin may hinder the unfolding of xyloglucan chains during cell deformation: implications of the mechanical performance of Arabidopsis hypocotyls with pectin alterations. Molecular Plant 2, 990–999. - PubMed
-
- Bashline L, Lei L, Li S, Gu Y. 2014. Cell wall, cytoskeleton, and cell expansion in higher plants. Molecular Plant 7, 586–600. - PubMed
-
- Baskin TI, Jensen OE. 2013. On the role of stress anisotropy in the growth of stems. Journal of Experimental Botany 64, 4697–4707. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

