Effects of Adaptation on Discrimination of Whisker Deflection Velocity and Angular Direction in a Model of the Barrel Cortex
- PMID: 29946250
- PMCID: PMC6006271
- DOI: 10.3389/fncom.2018.00045
Effects of Adaptation on Discrimination of Whisker Deflection Velocity and Angular Direction in a Model of the Barrel Cortex
Abstract
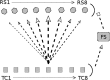
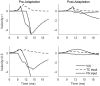
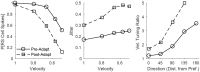
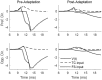
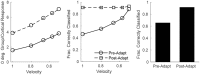
Two important stimulus features represented within the rodent barrel cortex are velocity and angular direction of whisker deflection. Each cortical barrel receives information from thalamocortical (TC) cells that relay information from a single whisker, and TC input is decoded by barrel regular-spiking (RS) cells through a feedforward inhibitory architecture (with inhibition delivered by cortical fast-spiking or FS cells). TC cells encode deflection velocity through population synchrony, while deflection direction is encoded through the distribution of spike counts across the TC population. Barrel RS cells encode both deflection direction and velocity with spike rate, and are divided into functional domains by direction preference. Following repetitive whisker stimulation, system adaptation causes a weakening of synaptic inputs to RS cells and diminishes RS cell spike responses, though evidence suggests that stimulus discrimination may improve following adaptation. In this work, I construct a model of the TC, FS, and RS cells comprising a single barrel system-the model incorporates realistic synaptic connectivity and dynamics and simulates both angular direction (through the spatial pattern of TC activation) and velocity (through synchrony of the TC population spikes) of a deflection of the primary whisker, and I use the model to examine direction and velocity selectivity of barrel RS cells before and after adaptation. I find that velocity and direction selectivity of individual RS cells (measured over multiple trials) sharpens following adaptation, but stimulus discrimination using a simple linear classifier by the RS population response during a single trial (a more biologically meaningful measure than single cell discrimination over multiple trials) exhibits strikingly different behavior-velocity discrimination is similar both before and after adaptation, while direction classification improves substantially following adaptation. This is the first model, to my knowledge, that simulates both whisker deflection velocity and angular direction and examines the ability of the RS population response to pinpoint both stimulus features within the context of adaptation.
Keywords: barrel cortex; deflection direction; deflection velocity; direction discrimination; feedforward inhibition; velocity discrimination; whisker.
Figures



Similar articles
-
Encoding whisker deflection velocity within the rodent barrel cortex using phase-delayed inhibition.J Comput Neurosci. 2014 Dec;37(3):387-401. doi: 10.1007/s10827-014-0535-3. Epub 2014 Oct 5. J Comput Neurosci. 2014. PMID: 25284340
-
Spiking and Excitatory/Inhibitory Input Dynamics of Barrel Cells in Response to Whisker Deflections of Varying Velocity and Angular Direction.Neuroscience. 2018 Jan 15;369:15-28. doi: 10.1016/j.neuroscience.2017.10.044. Epub 2017 Nov 6. Neuroscience. 2018. PMID: 29122591
-
Analysis of feedforward mechanisms of multiwhisker receptive field generation in a model of the rat barrel cortex.J Theor Biol. 2019 Sep 21;477:51-62. doi: 10.1016/j.jtbi.2019.06.008. Epub 2019 Jun 12. J Theor Biol. 2019. PMID: 31201881
-
Representation of tactile scenes in the rodent barrel cortex.Neuroscience. 2018 Jan 1;368:81-94. doi: 10.1016/j.neuroscience.2017.08.039. Epub 2017 Aug 23. Neuroscience. 2018. PMID: 28843997 Review.
-
Thalamocortical control of feed-forward inhibition in awake somatosensory 'barrel' cortex.Philos Trans R Soc Lond B Biol Sci. 2002 Dec 29;357(1428):1717-27. doi: 10.1098/rstb.2002.1156. Philos Trans R Soc Lond B Biol Sci. 2002. PMID: 12626006 Free PMC article. Review.
Cited by
-
A model of lateral interactions as the origin of multiwhisker receptive fields in rat barrel cortex.J Comput Neurosci. 2022 May;50(2):181-201. doi: 10.1007/s10827-021-00804-6. Epub 2021 Dec 1. J Comput Neurosci. 2022. PMID: 34854018
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

