SPG11 mutations cause widespread white matter and basal ganglia abnormalities, but restricted cortical damage
- PMID: 29946510
- PMCID: PMC6008284
- DOI: 10.1016/j.nicl.2018.05.031
SPG11 mutations cause widespread white matter and basal ganglia abnormalities, but restricted cortical damage
Abstract
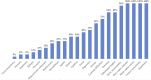
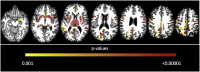
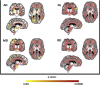
SPG11 mutations are the major cause of autosomal recessive Hereditary Spastic Paraplegia. The disease has a wide phenotypic variability indicating many regions of the nervous system besides the corticospinal tract are affected. Despite this, anatomical and phenotypic characterization is restricted. In the present study, we investigate the anatomical abnormalities related to SPG11 mutations and how they relate to clinical and cognitive measures. Moreover, we aim to depict how the disease course influences the regions affected, unraveling different susceptibility of specific neuronal populations. We performed clinical and paraclinical studies encompassing neuropsychological, neuroimaging, and neurophysiological tools in a cohort of twenty-five patients and age matched controls. We assessed cortical thickness (FreeSurfer software), deep grey matter volumes (T1-MultiAtlas tool), white matter microstructural damage (DTI-MultiAtlas) and spinal cord morphometry (Spineseg software) on a 3 T MRI scan. Mean age and disease duration were 29 and 13.2 years respectively. Sixty-four percent of the patients were wheelchair bound while 84% were demented. We were able to unfold a diffuse pattern of white matter integrity loss as well as basal ganglia and spinal cord atrophy. Such findings contrasted with a restricted pattern of cortical thinning (motor, limbic and parietal cortices). Electromyography revealed motor neuronopathy affecting 96% of the probands. Correlations with disease duration pointed towards a progressive degeneration of multiple grey matter structures and spinal cord, but not of the white matter. SPG11-related hereditary spastic paraplegia is characterized by selective neuronal vulnerability, in which a precocious and widespread white matter involvement is later followed by a restricted but clearly progressive grey matter degeneration.
Keywords: ACE-R, Addenbrooke's Cognitive Examination Revised; ALS, amyotrophic lateral sclerosis; CA, cord area; CE, cord eccentricity; CMAP, compound muscle action potential; CST, corticospinal tract; Complicated hereditary spastic paraplegia; DTI, diffusion tensor imaging; FA, fractional anisotropy; GM, grey matter; Grey matter; HSP, hereditary spastic paraplegia; LH, left hemisphere; MD, mean diffusivity; MOCA, Montreal cognitive assessment; Motor neuron disorder; NPI, neuropsychiatric inventory; PNP, sensory-motor polyneuropathy; PNS, peripheral nervous system; RH, right hemisphere; ROI, region of interest; SC, spinal cord; SNAP, sensory nerve action potential; SPG11; SPRS, Spastic Paraplegia Rating Scale; STS, cortex adjacent to the superior temporal sulcus; Spinal cord; Thinning of the corpus callosum; WES, whole exome sequencing; WM, white matter; White matter.
Figures
Similar articles
-
White and grey matter abnormalities in patients with SPG11 mutations.J Neurol Neurosurg Psychiatry. 2012 Aug;83(8):828-33. doi: 10.1136/jnnp-2011-300129. Epub 2012 Jun 13. J Neurol Neurosurg Psychiatry. 2012. PMID: 22696581
-
Diffusion tensor imaging in SPG11- and SPG4-linked hereditary spastic paraplegia.Int J Neurosci. 2014 Apr;124(4):261-70. doi: 10.3109/00207454.2013.836705. Epub 2013 Sep 27. Int J Neurosci. 2014. PMID: 23968121
-
Brain Damage and Gene Expression Across Hereditary Spastic Paraplegia Subtypes.Mov Disord. 2021 Jul;36(7):1644-1653. doi: 10.1002/mds.28519. Epub 2021 Feb 11. Mov Disord. 2021. PMID: 33576112
-
Hereditary spastic paraplegia type 11: Clinicogenetic lessons from 339 patients.J Clin Neurosci. 2021 Mar;85:67-71. doi: 10.1016/j.jocn.2020.11.036. Epub 2021 Jan 15. J Clin Neurosci. 2021. PMID: 33581793
-
Janus-faced spatacsin (SPG11): involvement in neurodevelopment and multisystem neurodegeneration.Brain. 2020 Aug 1;143(8):2369-2379. doi: 10.1093/brain/awaa099. Brain. 2020. PMID: 32355960 Free PMC article. Review.
Cited by
-
Multimodal MRI Longitudinal Assessment of White and Gray Matter in Different SPG Types of Hereditary Spastic Paraparesis.Front Neurosci. 2020 Jun 4;14:325. doi: 10.3389/fnins.2020.00325. eCollection 2020. Front Neurosci. 2020. PMID: 32581663 Free PMC article.
-
Recent advances in understanding hereditary spastic paraplegias and emerging therapies.Fac Rev. 2021 Mar 10;10:27. doi: 10.12703/r/10-27. eCollection 2021. Fac Rev. 2021. PMID: 33817696 Free PMC article. Review.
-
Cortical Damage Associated With Cognitive and Motor Impairment in Hereditary Spastic Paraplegia: Evidence of a Novel SPAST Mutation.Front Neurol. 2020 May 27;11:399. doi: 10.3389/fneur.2020.00399. eCollection 2020. Front Neurol. 2020. PMID: 32536902 Free PMC article.
-
Loss of the neural-specific BAF subunit ACTL6B relieves repression of early response genes and causes recessive autism.Proc Natl Acad Sci U S A. 2020 May 5;117(18):10055-10066. doi: 10.1073/pnas.1908238117. Epub 2020 Apr 20. Proc Natl Acad Sci U S A. 2020. PMID: 32312822 Free PMC article.
-
Spinal cord cross sign: a potential marker for hereditary spastic paraplegia type 5.Neuroradiology. 2025 Apr;67(4):1081-1090. doi: 10.1007/s00234-025-03543-y. Epub 2025 Jan 24. Neuroradiology. 2025. PMID: 39853345
References
-
- Anheim M., Lagier-Tourenne C., Stevanin G. SPG11 spastic paraplegia. A new cause of juvenile parkinsonism. J. Neurol. 2009;256:104–108. - PubMed
-
- Branco L.M., De Albuquerque M., De Andrade H.M., Bergo F.P., Nucci A., França M.C., Jr. Spinal cord atrophy correlates with disease duration and severity in amyotrophic lateral sclerosis. Amyotroph Lateral Scler. Frontotemporal Degener. 2014;15:93–97. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

