Comparison of Mesenchymal Stromal Cells Isolated from Murine Adipose Tissue and Bone Marrow in the Treatment of Spinal Cord Injury
- PMID: 29947256
- PMCID: PMC6158550
- DOI: 10.1177/0963689718780309
Comparison of Mesenchymal Stromal Cells Isolated from Murine Adipose Tissue and Bone Marrow in the Treatment of Spinal Cord Injury
Abstract
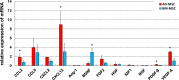
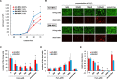
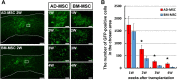
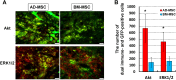
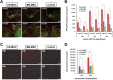
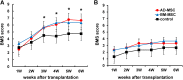
The use of mesenchymal stromal cell (MSC) transplantation to repair the injured spinal cord has shown consistent benefits in preclinical models. However, the low survival rate of grafted MSC is one of the most important problems. In the injured spinal cord, transplanted cells are exposed to hypoxic conditions and exposed to nutritional deficiency caused by poor vascular supply. Also, the transplanted MSCs face cytotoxic stressors that cause cell death. The aim of this study was to compare adipose-derived MSCs (AD-MSCs) and bone marrow-derived MSCs (BM-MSCs) isolated from individual C57BL6/J mice in relation to: (i) cellular characteristics, (ii) tolerance to hypoxia, oxidative stress and serum-free conditions, and (iii) cellular survival rates after transplantation. AD-MSCs and BM-MSCs exhibited a similar cell surface marker profile, but expressed different levels of growth factors and cytokines. To research their relative stress tolerance, both types of stromal cells were incubated at 20.5% O2 or 1.0% O2 for 7 days. Results showed that AD-MSCs were more proliferative with greater culture viability under these hypoxic conditions than BM-MSCs. The MSCs were also incubated under H2O2-induced oxidative stress and in serum-free culture medium to induce stress. AD-MSCs were better able to tolerate these stress conditions than BM-MSCs; similarly when transplanted into the spinal cord injury region in vivo, AD-MSCs demonstrated a higher survival rate post transplantation Furthermore, this increased AD-MSC survival post transplantation was associated with preservation of axons and enhanced vascularization, as delineated by increases in anti-gamma isotype of protein kinase C and CD31 immunoreactivity, compared with the BM-MSC transplanted group. Hence, our results indicate that AD-MSCs are an attractive alternative to BM-MSCs for the treatment of severe spinal cord injury. However, it should be noted that the motor function was equally improved following moderate spinal cord injury in both groups, but with no significant improvement seen unfortunately following severe spinal cord injury in either group.
Keywords: Adipose-derived mesenchymal stromal cell (AD-MSC); bone marrow-derived mesenchymal stromal cell (BM-MSC); spinal cord injury (SCI); treatment.
Conflict of interest statement
Figures
References
-
- Parr AM, Tator CH, Keating A. Bone marrow-derived mesenchymal stromal cells for the repair of central nervous system injury. Bone Marrow Transplant. 2007;40(7):609–619. - PubMed
-
- Lu Z, Xu S. ERK1/2 MAP kinases in cell survival and apoptosis. IUBMB Life. 2006;58(11):621–631. - PubMed
-
- Watanabe S, Uchida K, Nakajima H, Matsuo H, Sugita D, Yoshida A, Honjoh K, Johnson WE, Baba H. Early transplantation of mesenchymal stem cells after spinal cord injury relieves pain hypersensitivity through suppression of pain-related signaling cascades and reduced inflammatory cell recruitment. Stem Cells. 2015;33(6):1902–1914. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

