Connexin 43 Plays a Role in Pulmonary Vascular Reactivity in Mice
- PMID: 29954114
- PMCID: PMC6073802
- DOI: 10.3390/ijms19071891
Connexin 43 Plays a Role in Pulmonary Vascular Reactivity in Mice
Abstract
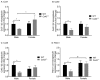
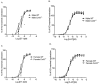
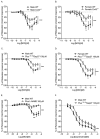
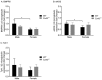
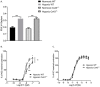
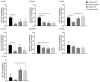
Pulmonary arterial hypertension (PAH) is a chronic condition characterized by vascular remodeling and increased vaso-reactivity. PAH is more common in females than in males (~3:1). Connexin (Cx)43 has been shown to be involved in cellular communication within the pulmonary vasculature. Therefore, we investigated the role of Cx43 in pulmonary vascular reactivity using Cx43 heterozygous (Cx43+/−) mice and 37,43Gap27, which is a pharmacological inhibitor of Cx37 and Cx43. Contraction and relaxation responses were studied in intra-lobar pulmonary arteries (IPAs) derived from normoxic mice and hypoxic mice using wire myography. IPAs from male Cx43+/− mice displayed a small but significant increase in the contractile response to endothelin-1 (but not 5-hydroxytryptamine) under both normoxic and hypoxic conditions. There was no difference in the contractile response to endothelin-1 (ET-1) or 5-hydroxytryptamine (5-HT) in IPAs derived from female Cx43+/−mice compared to wildtype mice. Relaxation responses to methacholine (MCh) were attenuated in IPAs from male and female Cx43+/− mice or by pre-incubation of IPAs with 37,43Gap27. Nω-Nitro-L-arginine methyl ester (l-NAME) fully inhibited MCh-induced relaxation. In conclusion, Cx43 is involved in nitric oxide (NO)-induced pulmonary vascular relaxation and plays a gender-specific and agonist-specific role in pulmonary vascular contractility. Therefore, reduced Cx43 signaling may contribute to pulmonary vascular dysfunction.
Keywords: connexin43 (Cx43); endothelin-1; gap junction; isoprenaline; nitric oxide; pulmonary arterial hypertension (PAH); serotonin; vascular reactivity.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Connexin 43 Affects Pulmonary Artery Reactivity via Changes in Nitric Oxide Production and Influences Proliferative and Migratory Responses in Mouse Pulmonary Artery Fibroblasts.Int J Mol Sci. 2025 Feb 1;26(3):1280. doi: 10.3390/ijms26031280. Int J Mol Sci. 2025. PMID: 39941047 Free PMC article.
-
Role of the gap junctions in the contractile response to agonists in pulmonary artery from two rat models of pulmonary hypertension.Respir Res. 2011 Mar 17;12(1):30. doi: 10.1186/1465-9921-12-30. Respir Res. 2011. PMID: 21414209 Free PMC article.
-
Down-regulation of connexin 43 contributes to structure and function of pulmonary artery in nicotine-administered mice.Toxicol Lett. 2023 Mar 15;377:1-13. doi: 10.1016/j.toxlet.2023.01.008. Epub 2023 Jan 28. Toxicol Lett. 2023. PMID: 36720419
-
Connexin 43 mediates endothelium-derived hyperpolarizing factor-induced vasodilatation in subcutaneous resistance arteries from healthy pregnant women.Am J Physiol Heart Circ Physiol. 2007 Feb;292(2):H1026-32. doi: 10.1152/ajpheart.00797.2006. Epub 2006 Nov 3. Am J Physiol Heart Circ Physiol. 2007. PMID: 17085540
-
Importance of Cx43 for Right Ventricular Function.Int J Mol Sci. 2021 Jan 20;22(3):987. doi: 10.3390/ijms22030987. Int J Mol Sci. 2021. PMID: 33498172 Free PMC article. Review.
Cited by
-
New Insights into Pulmonary Hypertension: A Role for Connexin-Mediated Signalling.Int J Mol Sci. 2021 Dec 29;23(1):379. doi: 10.3390/ijms23010379. Int J Mol Sci. 2021. PMID: 35008804 Free PMC article. Review.
-
Chronic Hypoxia Decreases Endothelial Connexin 40, Attenuates Endothelium-Dependent Hyperpolarization-Mediated Relaxation in Small Distal Pulmonary Arteries, and Leads to Pulmonary Hypertension.J Am Heart Assoc. 2020 Dec 15;9(24):e018327. doi: 10.1161/JAHA.120.018327. Epub 2020 Dec 12. J Am Heart Assoc. 2020. PMID: 33307937 Free PMC article.
-
An Overview of the Focus of the International Gap Junction Conference 2017 and Future Perspectives.Int J Mol Sci. 2018 Sep 18;19(9):2823. doi: 10.3390/ijms19092823. Int J Mol Sci. 2018. PMID: 30231591 Free PMC article.
-
HIF-1α promotes the proliferation and migration of pulmonary arterial smooth muscle cells via activation of Cx43.J Cell Mol Med. 2021 Nov;25(22):10663-10673. doi: 10.1111/jcmm.17003. Epub 2021 Oct 26. J Cell Mol Med. 2021. PMID: 34698450 Free PMC article.
-
Connexin 43 plays a role in proliferation and migration of pulmonary arterial fibroblasts in response to hypoxia.Pulm Circ. 2020 Jul 6;10(3):2045894020937134. doi: 10.1177/2045894020937134. eCollection 2020 Jul-Sep. Pulm Circ. 2020. PMID: 32670564 Free PMC article.
References
-
- Eddahibi S., Guignabert C., Barlier-Mur A.M., Dewachter L., Fadel E., Dartevelle P., Humbert M., Simonneau G., Hanoun N., Saurini F., et al. Cross Talk between Endothelial and Smooth Muscle Cells in Pulmonary Hypertension: Critical Role for Serotonin-Induced Smooth Muscle Hyperplasia. Circulation. 2006;113:1857–1864. doi: 10.1161/CIRCULATIONAHA.105.591321. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous

