Solid-State Insight Into the Action of a Pharmaceutical Solvate: Structural, Thermal, and Dissolution Analysis of Indinavir Sulfate Ethanolate
- PMID: 29960024
- PMCID: PMC6170162
- DOI: 10.1016/j.xphs.2018.06.020
Solid-State Insight Into the Action of a Pharmaceutical Solvate: Structural, Thermal, and Dissolution Analysis of Indinavir Sulfate Ethanolate
Abstract
The crystal structure of indinavir sulfate, a pharmaceutical administered as an ethanol solvate, is presented, revealing a unique channel/ionic solvate structure to be characteristic of the compound. The properties of the material with regard to thermal treatment and water adsorption follow closely from the structure. The in situ amorphization of the pharmaceutical upon contacting liquid water is observed and highlights the unique dissolution enhancement of marketing the crystalline solvate dosage. Through survey of published crystal structures, an ambiguous sulfate/bisulfate ionization state is also observed in the crystal, which challenges the general understanding of the pharmaceutical. This study provides a solid-state insight into the function of a special multicomponent crystalline pharmaceutical form.
Keywords: amorphism; crystal structure; desolvation; salts; solvates.
Copyright © 2018 American Pharmacists Association®. Published by Elsevier Inc. All rights reserved.
Figures


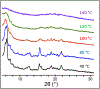
Similar articles
-
Desolvation behavior of indinavir sulfate ethanol and follow-up by terahertz spectroscopy.Int J Pharm. 2019 Aug 15;567:118446. doi: 10.1016/j.ijpharm.2019.06.037. Epub 2019 Jun 18. Int J Pharm. 2019. PMID: 31220565
-
Solid state characterization of the anti-HIV drug TMC114: interconversion of amorphous TMC114, TMC114 ethanolate and hydrate.Eur J Pharm Sci. 2009 Dec 8;38(5):489-97. doi: 10.1016/j.ejps.2009.09.013. Epub 2009 Sep 24. Eur J Pharm Sci. 2009. PMID: 19782132
-
Influence of solvents on the variety of crystalline forms of erythromycin.AAPS PharmSci. 2003;5(2):E12. doi: 10.1208/ps050212. AAPS PharmSci. 2003. PMID: 12866939 Free PMC article.
-
High-throughput crystallization: polymorphs, salts, co-crystals and solvates of pharmaceutical solids.Adv Drug Deliv Rev. 2004 Feb 23;56(3):275-300. doi: 10.1016/j.addr.2003.10.020. Adv Drug Deliv Rev. 2004. PMID: 14962582 Review.
-
Impact of solid state properties on developability assessment of drug candidates.Adv Drug Deliv Rev. 2004 Feb 23;56(3):321-34. doi: 10.1016/j.addr.2003.10.007. Adv Drug Deliv Rev. 2004. PMID: 14962584 Review.
Cited by
-
Desolvation Processes in Channel Solvates of Niclosamide.Mol Pharm. 2023 Nov 6;20(11):5554-5562. doi: 10.1021/acs.molpharmaceut.3c00481. Epub 2023 Oct 18. Mol Pharm. 2023. PMID: 37850910 Free PMC article.
-
Far-Infrared Spectroscopy as a Probe for Polymorph Discrimination.J Pharm Sci. 2019 May;108(5):1915-1920. doi: 10.1016/j.xphs.2018.12.020. Epub 2018 Dec 30. J Pharm Sci. 2019. PMID: 30599167 Free PMC article.
References
-
- Hilfiker R, Blatter F, Raumer MV. Relevance of Solid-state Properties for Pharmaceutical Products In: Hilfiker R, eds. Polymorphism: in the Pharmaceutical Industry. Weinheim: Wiley-VCH Verlag GmbH & Co. KGaA.; 2006:1–19.
-
- Brittain HG. Theory and Origin of Polymorphism In: Brittain HG, eds. Polymorphism in Pharmaceutical Solids. New York: Marcel Dekker.; 1999:1–31.
-
- Griesser UJ. Relevance of Solid-state Properties for Pharmaceutical Products In: Hilfiker R, eds. Polymorphism: in the Pharmaceutical Industry. Weinheim: Wiley-VCH Verlag GmbH & Co. KGaA.; 2006:211–233.
-
- Douillet J, Stevenson N, Lee M, Mallet F, Ward R, Aspin P, Dennehy DR, Camus L. Development of a solvate as an active pharmaceutical ingredient: Developability, crystallisation and isolation challenges. J. Cryst. Growth 2012;342(1):2–8.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

