Alternative splicing of (ppp1r12a/mypt1) in zebrafish produces a novel myosin phosphatase targeting subunit
- PMID: 29960069
- PMCID: PMC6123272
- DOI: 10.1016/j.gene.2018.06.092
Alternative splicing of (ppp1r12a/mypt1) in zebrafish produces a novel myosin phosphatase targeting subunit
Abstract
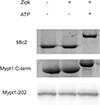
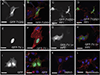
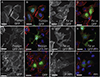
Myosin phosphatase is an evolutionarily conserved regulator of actomyosin contractility, comprised of a regulatory subunit (Mypt1), and a catalytic subunit (PP1). Zebrafish has become an ideal model organism for the study of the genetic and cell physiological role of the myosin phosphatase in morphogenesis and embryonic development. We identified and characterized a novel splice variant of Mypt1 (ppp1r12a-tv202) from zebrafish, which is widely expressed during early embryonic development. Importantly, mutant alleles and antisense morpholinos that have been used to demonstrate the important role of Mypt1 in early development, not only disrupt the longer splice variants, but also tv202. The protein product of ppp1r12a-tv202 (Mypt1-202) contains the PP1-binding N-terminus, but lacks the regulatory C-terminus, which contains two highly conserved inhibitory phosphorylation sites. We observed that the protein product of tv202 assembled a constitutively active myosin phosphatase uninhibited by kinases such as Zipk. Thus, we propose that Mypt1-202 plays an important role in maintaining baseline Mlc2 dephosphorylation and actomyosin relaxation during early zebrafish development.
Keywords: Actomyosin; Convergent extension; Gastrulation; Myosin phosphatase; Mypt1; PP1; Splicing; Zebrafish.
Copyright © 2018 Elsevier B.V. All rights reserved.
Conflict of interest statement
Conflict of interest
The authors declare no conflict of interest
Figures



Similar articles
-
Protein phosphatase 1 β paralogs encode the zebrafish myosin phosphatase catalytic subunit.PLoS One. 2013 Sep 11;8(9):e75766. doi: 10.1371/journal.pone.0075766. eCollection 2013. PLoS One. 2013. PMID: 24040418 Free PMC article.
-
Physiological vs. pharmacological signalling to myosin phosphorylation in airway smooth muscle.J Physiol. 2017 Oct 1;595(19):6231-6247. doi: 10.1113/JP274715. Epub 2017 Aug 24. J Physiol. 2017. PMID: 28749013 Free PMC article.
-
Myosin phosphatase targeting subunit 1 affects cell migration by regulating myosin phosphorylation and actin assembly.Exp Cell Res. 2005 Apr 1;304(2):506-17. doi: 10.1016/j.yexcr.2004.11.025. Epub 2004 Dec 30. Exp Cell Res. 2005. PMID: 15748895
-
Myosin phosphatase: Unexpected functions of a long-known enzyme.Biochim Biophys Acta Mol Cell Res. 2019 Jan;1866(1):2-15. doi: 10.1016/j.bbamcr.2018.07.023. Epub 2018 Aug 2. Biochim Biophys Acta Mol Cell Res. 2019. PMID: 30076859 Review.
-
Regulation of Smooth Muscle Myosin Light Chain Phosphatase by Multisite Phosphorylation of the Myosin Targeting Subunit, MYPT1.Cardiovasc Hematol Disord Drug Targets. 2018;18(1):4-13. doi: 10.2174/1871529X18666180326120638. Cardiovasc Hematol Disord Drug Targets. 2018. PMID: 29577868 Review.
Cited by
-
The Evolution of Duplicated Genes of the Cpi-17/Phi-1 (ppp1r14) Family of Protein Phosphatase 1 Inhibitors in Teleosts.Int J Mol Sci. 2020 Aug 9;21(16):5709. doi: 10.3390/ijms21165709. Int J Mol Sci. 2020. PMID: 32784920 Free PMC article.
-
Clinical report and genetic analysis of a neonate with genitourinary and/or brain malformation syndrome caused by a non-coding sequence variant of PPP1R12A.Mol Genet Genomic Med. 2023 Oct;11(10):e2223. doi: 10.1002/mgg3.2223. Epub 2023 Jun 5. Mol Genet Genomic Med. 2023. PMID: 37272772 Free PMC article.
References
-
- Hirano K, Derkach DN, Hirano M, Nishimura J & Kanaide H Protein kinase network in the regulation of phosphorylation and dephosphorylation of smooth muscle myosin light chain. Molecular and Cellular Biochemistry 248, 105–114(2003). - PubMed
-
- Grassie ME, Moffat LD, Walsh MP & MacDonald JA The myosin phosphatase targeting protein (MYPT) family: A regulated mechanism for achieving substrate specificity of the catalytic subunit of protein phosphatase type 1δ. Archives of Biochemistry and Biophysics 510, 147–159 (2011). - PubMed
-
- Ito M, Nakano T, Erdodi F & Hartshome DJ Myosin phosphatase: Structure, regulation and function. Molecular and Cellular Biochemistry 259, 197–209 (2004). - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

